Pre-scriptum (dated 26 June 2020): My views on the true nature of light and matter have evolved significantly as part of my explorations of a more realist (classical) explanation of quantum mechanics. If you are reading this, then you are probably looking for not-to-difficult reading. In that case, I would suggest you read my re-write of Feynman’s introductory lecture to QM. If you want something shorter, you can also read my paper on what I believe to be the true Principles of Physics.
Original post:
In my previous post, I wrote that I was puzzled by that relation between the energy and the size of a particle: higher-energy photons are supposed to be smaller and, pushing that logic to the limit, we get photons becoming black holes at the Planck scale. Now, understanding what the Planck scale is all about, is important to understand why we’d need a GUT, and so I do want to explore that relation between size and energy somewhat further.
I found the answer by a coincidence. We’ll call it serendipity. 🙂 Indeed, an acquaintance of mine who is very well versed in physics pointed out a terrible mistake in (some of) my reasoning in the previous posts: photons do not have a de Broglie wavelength. They just have a wavelength. Full stop. It immediately reduced my bemusement about that energy-size relation and, in the end, eliminated it completely. So let’s analyze that mistake – which seems to be a fairly common freshman mistake judging from what’s being written about it in some of the online discussions on physics.
If photons are not to be associated with a de Broglie wave, it basically means that the Planck relation has nothing to do with the de Broglie relation, even if these two relations are identical from a pure mathematical point of view:
- The Planck relation E = hν states that electromagnetic waves with frequency ν are a bunch of discrete packets of energy referred to as photons, and that the energy of these photons is proportional to the frequency of the electromagnetic wave, with the Planck constant h as the factor of proportionality. In other words, the natural unit to measure their energy is h, which is why h is referred to as the quantum of action.
- The de Broglie relation E = hf assigns a de Broglie wave with frequency f to a matter particle with energy E = mc2 = γm0c2. [The factor γ in this formula is the Lorentz factor: γ = (1 – v2/c2)–1/2. It just corrects for the relativistic effect on mass as the velocity of the particle (v) gets closer to the speed of light (c).]
These are two very different things: photons do not have rest mass (which is why they can travel at light speed) and, hence, they are not to be considered as matter particles. Therefore, one should not assign a de Broglie wave to them. So what are they then? A photon is a wave packet but it’s an electromagnetic wave packet. Hence, its wave function is not some complex-valued psi function Ψ(x, t). What is oscillating in the illustration below (let’s say this is a procession of photons) is the electric field vector E. [To get the full picture of the electromagnetic wave, you should also imagine a (tiny) magnetic field vector B, which oscillates perpendicular to E), but that does not make much of a difference. Finally, in case you wonder about these dots: the red and green dot just make it clear that phase and group velocity of the wave are the same: vg = vp = v = c.]  The point to note is that we have a real wave here: it is not a de Broglie wave. A de Broglie wave is a complex-valued function Ψ(x, t) with two oscillating parts: (i) the so-called real part of the complex value Ψ, and (ii) the so-called imaginary part (and, despite its name, that counts as much as the real part when working with Ψ !). That’s what’s shown in the examples of complex (standing) waves below: the blue part is one part (let’s say the real part), and then the salmon color is the other part. We need to square the modulus of that complex value to find the probability P of detecting that particle in space at point x at time t: P(x, t) = |Ψ(x, t)|2. Now, if we would write Ψ(x, t) as Ψ = u(x, t) + iv(x, t), then u(x, t) is the real part, and v(x, t) is the imaginary part. |Ψ(x, t)|2 is then equal to u2 + u2 so that shows that both the blue as well as the salmon amplitude matter when doing the math.
The point to note is that we have a real wave here: it is not a de Broglie wave. A de Broglie wave is a complex-valued function Ψ(x, t) with two oscillating parts: (i) the so-called real part of the complex value Ψ, and (ii) the so-called imaginary part (and, despite its name, that counts as much as the real part when working with Ψ !). That’s what’s shown in the examples of complex (standing) waves below: the blue part is one part (let’s say the real part), and then the salmon color is the other part. We need to square the modulus of that complex value to find the probability P of detecting that particle in space at point x at time t: P(x, t) = |Ψ(x, t)|2. Now, if we would write Ψ(x, t) as Ψ = u(x, t) + iv(x, t), then u(x, t) is the real part, and v(x, t) is the imaginary part. |Ψ(x, t)|2 is then equal to u2 + u2 so that shows that both the blue as well as the salmon amplitude matter when doing the math.
So, while I may have given the impression that the Planck relation was like a limit of the de Broglie relation for particles with zero rest mass traveling at speed c, that’s just plain wrong ! The description of a particle with zero rest mass fits a photon but the Planck relation is not the limit of the de Broglie relation: photons are photons, and electrons are electrons, and an electron wave has nothing to do with a photon. Electrons are matter particles (fermions as physicists would say), and photons are bosons, i.e. force carriers.
Let’s now re-examine the relationship between the size and the energy of a photon. If the wave packet below would represent an (ideal) photon, what is its energy E as a function of the electric and magnetic field vectors E and B? [Note that the (non-boldface) E stands for energy (i.e. a scalar quantity, so it’s just a number) indeed, while the (italic and bold) E stands for the (electric) field vector (so that’s something with a magnitude (E – with the symbol in italics once again to distinguish it from energy E) and a direction).] Indeed, if a photon is nothing but a disturbance of the electromagnetic field, then the energy E of this disturbance – which obviously depends on E and B – must also be equal to E = hν according to the Planck relation. Can we show that?
Well… Let’s take a snapshot of a plane-wave photon, i.e. a photon oscillating in a two-dimensional plane only. That plane is perpendicular to our line of sight here:

Because it’s a snapshot (time is not a variable), we may look at this as an electrostatic field: all points in the interval Δx are associated with some magnitude E (i.e. the magnitude of our electric field E), and points outside of that interval have zero amplitude. It can then be shown (just browse through any course on electromagnetism) that the energy density (i.e. the energy per unit volume) is equal to (1/2)ε0E2 (ε0 is the electric constant which we encountered in previous posts already). To calculate the total energy of this photon, we should integrate over the whole distance Δx, from left to right. However, rather than bothering you with integrals, I think that (i) the ε0E2/2 formula and (ii) the illustration above should be sufficient to convince you that:
- The energy of a photon is proportional to the square of the amplitude of the electric field. Such E ∝ A2 relation is typical of any real wave, be they water waves or electromagnetic waves. So if we would double, triple, or quadruple its amplitude (i.e. the magnitude E of the electric field E), then the energy of this photon with be multiplied with four, nine times and sixteen respectively.
- If we would not change the amplitude of the wave above but double, triple or quadruple its frequency, then we would only double, triple or quadruple its energy: there’s no exponential relation here. In other words, the Planck relation E = hν makes perfect sense, because it reflects that simple proportionality: there is nothing to be squared.
- If we double the frequency but leave the amplitude unchanged, then we can imagine a photon with the same energy occupying only half of the Δx space. In fact, because we also have that universal relationship between frequency and wavelength (the propagation speed of a wave equals the product of its wavelength and its frequency: v = λf), we would have to halve the wavelength (and, hence, that would amount to dividing the Δx by two) to make sure our photon is still traveling at the speed of light.
Now, the Planck relation only says that higher energy is associated with higher frequencies: it does not say anything about amplitudes. As mentioned above, if we leave amplitudes unchanged, then the same Δx space will accommodate a photon with twice the frequency and twice the energy. However, if we would double both frequency and amplitude, then the photon would occupy only half of the Δx space, and still have twice as much energy. So the only thing I now need to prove is that higher-frequency electromagnetic waves are associated with larger-amplitude E‘s. Now, while that is something that we get straight out of the the laws of electromagnetic radiation: electromagnetic radiation is caused by oscillating electric charges, and it’s the magnitude of the acceleration (written as a in the formula below) of the oscillating charge that determines the amplitude. Indeed, for a full write-up of these ‘laws’, I’ll refer to a textbook (or just download Feynman’s 28th Lecture on Physics), but let me just give the formula for the (vertical) component of E: 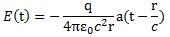
You will recognize all of the variables and constants in this one: the electric constant ε0, the distance r, the speed of light (and our wave) c, etcetera. The ‘a’ is the acceleration: note that it’s a function not of t but of (t – r/c), and so we’re talking the so-called retarded acceleration here, but don’t worry about that.
Now, higher frequencies effectively imply a higher magnitude of the acceleration vector, and so that’s what’s I had to prove and so we’re done: higher-energy photons not only have higher frequency but also larger amplitude, and so they take less space.
It would be nice if I could derive some kind of equation to specify the relation between energy and size, but I am not that advanced in math (yet). 🙂 I am sure it will come.
Post scriptum 1: The ‘mistake’ I made obviously fully explains why Feynman is only interested in the amplitude of a photon to go from point A to B, and not in the amplitude of a photon to be at point x at time t. The question of the ‘size of the arrows’ then becomes a question related to the so-called propagator function, which gives the probability amplitude for a particle (a photon in this case) to travel from one place to another in a given time. The answer seems to involve another important buzzword when studying quantum mechanics: the gauge parameter. However, that’s also advanced math which I don’t master (as yet). I’ll come back on it… Hopefully… 🙂
Post scriptum 2: As I am re-reading some of my post now (i.e. on 12 January 2015), I noted how immature this post is. I wanted to delete it, but finally I didn’t, as it does illustrate my (limited) progress. I am still struggling with the question of a de Broglie wave for a photon, but I dare to think that my analysis of the question at least is a bit more mature now: please see one of my other posts on it.
Some content on this page was disabled on June 20, 2020 as a result of a DMCA takedown notice from Michael A. Gottlieb, Rudolf Pfeiffer, and The California Institute of Technology. You can learn more about the DMCA here:

