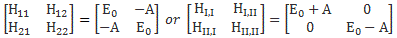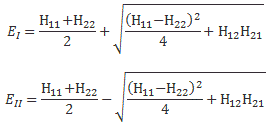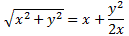In my post on the hydrogen atom, I explained its stability using the following graph out of Feynman’s Lectures. It shows an equilibrium state for the H2 molecule with an energy level that’s about 5 eV (ΔE/EH ≈ −0.375 ⇔ ΔE ≈ −0.375×13.6 eV = 5.1 eV) lower than the energy of two separate hydrogen atoms (2H).
 The lower energy level is denoted by EII and refers to a state, which we also denoted as state II, that’s associated with some kind of molecular orbital for both electrons, resulting in more (shared) space where the two electrons can have a low potential energy, as Feynman puts it, so “the electron can spread out—lowering its kinetic energy—without increasing its potential energy.” The electrons have opposite spin. The have to have opposite spin because our formula for state II would violate the Fermi exclusion principle if they would not have opposite spin. Indeed, if the two electrons would not have opposite spin, the formula for our CII amplitude, would be violating the rule that, when identical fermions are involved, and we’re adding amplitudes, then we should do so with a negative sign for the exchanged case. So our CII = 〈II|ψ〉 = (1/√2)[〈1|ψ〉 + 〈2|ψ〉] = (1/√2)[〈2|ψ〉 + 〈1|ψ〉] would be problematic: when we switch the electrons, we should get a minus sign.
The lower energy level is denoted by EII and refers to a state, which we also denoted as state II, that’s associated with some kind of molecular orbital for both electrons, resulting in more (shared) space where the two electrons can have a low potential energy, as Feynman puts it, so “the electron can spread out—lowering its kinetic energy—without increasing its potential energy.” The electrons have opposite spin. The have to have opposite spin because our formula for state II would violate the Fermi exclusion principle if they would not have opposite spin. Indeed, if the two electrons would not have opposite spin, the formula for our CII amplitude, would be violating the rule that, when identical fermions are involved, and we’re adding amplitudes, then we should do so with a negative sign for the exchanged case. So our CII = 〈II|ψ〉 = (1/√2)[〈1|ψ〉 + 〈2|ψ〉] = (1/√2)[〈2|ψ〉 + 〈1|ψ〉] would be problematic: when we switch the electrons, we should get a minus sign.
We do get that minus sign for state I:
〈I|ψ〉 = (1/√2)[〈1|ψ〉 − 〈2|ψ〉] = −(1/√2)[〈2|ψ〉 − 〈1|ψ〉]
To make a long story short, state II is the equilibrium state, and so that’s an H2 molecule with two electrons with opposite spins that share a molecular orbital, rather than moving around in some atomic orbital.
The question is: can we generalize this analysis? I mean… We’ve spent a lot of time so as to make sure we understand this one particular case. What’s the use of such analysis if we can’t generalize? We shouldn’t be doing nitty-gritty all of the time, isn’t it?
You’re right. The thing is: we can easily generalize. We’ve learned to play with those Hamiltonian matrices now, and so let’s do the ‘same-same but different’ with other systems. Let’s replace one of the two protons in the two-protons-one-electron model by a much heavier ion—say, lithium. [The example is not random, of course: lithium is very easily ionized, which is why it’s used in batteries.]
We need to think of the Hamiltonian again, right? We’re now in a situation in which the Hamiltonian coefficients H11 and H22 are likely to be different. We’ve lost the symmetry: if the electron is near the lithium ion, then we can’t assume the system has the same energy as when it’s near the hydrogen nucleus (in case you forgot: that’s what the proton is, really). Because we’ve lost the symmetry, we no longer have these ‘easy’ Hamiltonians:
We need to look at the original formulas for E1 and E2 once again. Let me write them down:
Of course, H12 and H21 will still be equal to A, and so… Well… Let me simplify my life and copy Feynman:
There’s several things here. First, note that approximation to the square root:
We’re only allowed to do that if y is much smaller than x, with x = 1 and y = 2A/(H11 − H22). In fact, the condition is usually written as 0 ≤ y/x ≤ 1/2, so we take the A/(H11 − H22) ratio as (much) less than one, indeed. So the second term in the energy difference EI − EII = (H11 − H22) + 2A·A/(H11 − H22) is surely smaller than 2A. But there’s the first term, of course: H11 − H22. However, that’s there anyway, and so we should actually be looking at the additional separation, so that’s where the A comes in, and so that’s the second term: 2A·A/(H11 − H22) which, as mentioned, is smaller by the factor A/(H11 − H22), which is less than one. So Feynman’s conclusion is correct: “The binding of unsymmetric diatomic molecules is generally very weak.“
However, that’s not the case when binding two ions by two electrons, which is referred to as a two-electron binding, which is the most common valence bond. Let me simplify my life once more and quote once again:
What he’s saying is that H11 and H22 are one and the same once again, and equal to E0, because both ions can take one electron, so there’s no difference between state 1 and state 2 in that regard. So the energy difference is 2A once more and we’ve got good covalent binding. [Note that the term ‘covalent’ just refers to sharing electrons, so their value is shared, so to speak.]
Now, this result is, of course, subject to the hypothesis that the electron is more or less equally attracted to both ions, which may or may not be the case. If it’s not the case, we’ll have what’s referred to as ‘ionic’ binding. Again, I’ll let Feynman explain it, as it’s pretty straightforward and so it’s no use to try to write another summary of this:
So… That’s it, really. As Feynman puts it, by way of conclusion: “You can now begin to see how it is that many of the facts of chemistry can be most clearly understood in terms of a quantum mechanical description.”
Most clearly? Well… I guess that, at the very least, we’re “beginning to see” something here, aren’t we? 🙂
Some content on this page was disabled on June 16, 2020 as a result of a DMCA takedown notice from The California Institute of Technology. You can learn more about the DMCA here:
https://wordpress.com/support/copyright-and-the-dmca/
Some content on this page was disabled on June 16, 2020 as a result of a DMCA takedown notice from The California Institute of Technology. You can learn more about the DMCA here:https://wordpress.com/support/copyright-and-the-dmca/
Some content on this page was disabled on June 16, 2020 as a result of a DMCA takedown notice from The California Institute of Technology. You can learn more about the DMCA here: