On 25 September 1933, Paul Ehrenfest took his son Wassily, who was suffering from Down syndrome, for a walk in the park. He shot him, and then killed himself. He was only 53. That’s my age bracket. From the letters he left (here is a summary in Dutch), we know his frustration of not being able to arrive at some kind of common-sense interpretation of the new quantum physics played a major role in the anxiety that had brought him to this point. He had taken courses from Ludwig Boltzmann as an aspiring young man. We, therefore, think Boltzmann’s suicide – for similar reasons – might have troubled him too.
His suicide did not come unexpectedly: he had announced it. In one of his letters to Einstein, he complains about ‘indigestion’ from the ‘unendlicher Heisenberg-Born-Dirac-Schrödinger Wurstmachinen-Physik-Betrieb.’ I’ll let you google-translate that. He also seems to have gone through the trouble of summarizing all his questions on the new approach in an article in what was then one of the top journals for physics: Einige die Quantenmechanik betreffende Erkundigungsfrage, Zeitschrift für Physik 78 (1932) 555-559 (quoted in the above-mentioned review article). This I’ll translate: Some Questions about Quantum Mechanics.

Paul Ehrenfest in happier times (painting by Harm Kamerlingh Onnes in 1920)
A diplomat-friend of mine once remarked this: “It is good you are studying physics only as a pastime. Professional physicists are often troubled people—miserable.” It is an interesting observation from a highly intelligent outsider. To be frank, I understand this strange need to probe things at the deepest level—to be able to explain what might or might not be the case (I am using Wittgenstein’s definition of reality here). Even H.A. Lorentz, who – fortunately, perhaps – died before his successor did what he did, was becoming quite alarmist about the sorry state of academic physics near the end of his life—and he, Albert Einstein, and so many others were not alone. Not then, and not now. All of the founding fathers of quantum mechanics ended up becoming pretty skeptical about the theory they had created. We have documented that elsewhere so we won’t talk too much about it here. Even John Stewart Bell himself – one of the third generation of quantum physicists, we may say – did not like his own ‘No Go Theorem’ and thought that some “radical conceptual renewal”[1] might disprove his conclusions.
The Born-Heisenberg revolution has failed: most – if not all – of contemporary high-brow physicist are pursuing alternative theories—in spite, or because, of the academic straitjackets they have to wear. If a genius like Ehrenfest didn’t buy it, then I won’t buy it either. Furthermore, the masses surely don’t buy it and, yes, truth – in this domain too – is, fortunately, being defined more democratically nowadays. The Nobel Prize Committee will have to do some serious soul-searching—if not five years from now, then ten.
We feel sad for the physicists who died unhappily—and surely for those who took their life out of depression—because the common-sense interpretation they were seeking is so self-evident: de Broglie’s intuition in regard to matter being wavelike was correct. He just misinterpreted its nature: it is not a linear but a circular wave. We quickly insert the quintessential illustration (courtesy of Celani, Vassallo and Di Tommaso) but we refer the reader for more detail to our articles or – more accessible, perhaps – our manuscript for the general public.

The equations are easy. The mass of an electron – any matter-particle, really – is the equivalent mass of the oscillation of the charge it carries. This oscillation is, most probably, statistically regular only. So we think it’s chaotic, actually, but we also think the words spoken by Lord Pollonius in Shakespeare’s Hamlet apply to it: “Though this be madness, yet there is method in ‘t.” This means we can meaningfully speak of a cycle time and, therefore, of a frequency. Erwin Schrödinger stumbled upon this motion while exploring solutions to Dirac’s wave equation for free electrons, and Dirac immediately grasped the significance of Schrödinger’s discovery, because he mentions Schrödinger’s discovery rather prominently in his Nobel Prize Lecture:
“It is found that an electron which seems to us to be moving slowly, must actually have a very high frequency oscillatory motion of small amplitude superposed on the regular motion which appears to us. As a result of this oscillatory motion, the velocity of the electron at any time equals the velocity of light. This is a prediction which cannot be directly verified by experiment, since the frequency of the oscillatory motion is so high and its amplitude is so small. But one must believe in this consequence of the theory, since other consequences of the theory which are inseparably bound up with this one, such as the law of scattering of light by an electron, are confirmed by experiment.” (Paul A.M. Dirac, Theory of Electrons and Positrons, Nobel Lecture, December 12, 1933)
Unfortunately, Dirac confuses the concept of the electron as a particle with the concept of the (naked) charge inside. Indeed, the idea of an elementary (matter-)particle must combine the idea of a charge and its motion to account for both the particle- as well as the wave-like character of matter-particles. We do not want to dwell on all of this because we’ve written too many papers on this already. We just thought it would be good to sum up the core of our common-sense interpretation of physics. Why? To honor Boltzmann and Ehrenfest: I think of their demise as a sacrifice in search for truth.
[…]
OK. That sounds rather tragic—sorry for that! For the sake of brevity, we will just describe the electron here.
I. Planck’s quantum of action (h) and the speed of light (c) are Nature’s most fundamental constants. Planck’s quantum of action relates the energy of a particle to its cycle time and, therefore, to its frequency:
(1) h = E·T = E/f ⇔ ħ = E/ω
The charge that is whizzing around inside of the electron has zero rest mass, and so it whizzes around at the speed of light: the slightest force on it gives it an infinite acceleration. It, therefore, acquires a relativistic mass which is equal to mγ = me/2 (we refer to our paper(s) for a relativistically correct geometric argument). The momentum of the pointlike charge, in its circular or orbital motion, is, therefore, equal to p = mγ·c = me·c/2.
The (angular) frequency of the oscillation is also given by the formula for the (angular) velocity:
(2) c = a·ω ⇔ ω = c/a
While Eq. (1) is a fundamental law of Nature, Eq. (2) is a simple geometric or mathematical relation only.
II. From (1) and (2), we can now calculate the radius of this tiny circular motion as:
(3a) ħ = E/ω = E·a/c ⇔ a = (ħ·c)/E
Because we know the mass of the electron is the inertial mass of the state of motion of the pointlike charge, we may use Einstein’s mass-energy equivalence relation to rewrite this as the Compton radius of the electron:
(3b) a = (ħ·c)/E = (ħ·c)/(me·c2) = ħ/(me·c)
Note that we only used two fundamental laws of Nature so far: the Planck-Einstein relation and Einstein’s mass-energy equivalence relation.
III. We must also be able to express the Planck-Einstein quantum as the product of the momentum (p) of the pointlike charge and some length λ:
(4) h = p·λ
The question here is: what length? The circumference of the loop, or its radius? The same geometric argument we used to derive the effective mass of the pointlike charge as it whizzes around at lightspeed around its center, tells us the centripetal force acts over a distance that is equal to two times the radius. Indeed, the relevant formula for the centripetal force is this:
(5) F = (mγ/me)·(E/a) = E/2a
We can therefore reduce Eq. (4) by dividing it by 2π. We then get reduced, angular or circular (as opposed to linear) concepts:
(6) ħ = (p·λ)/(2π) = (me·c/2)·(λ/π) = (me·c/2)·(2a) = me·c·a ⇔ ħ/a = me·c
We can verify the logic of our reasoning by substituting a for the Compton radius:
ħ = p·λ = me·c·a = me·c·a = me·c·ħ/(me·c) = ħ
IV. We can, finally, re-confirm the logic of our reason by re-deriving Einstein’s mass-energy equivalence relation as well as the Planck-Einstein relation using the ω = c/a and the ħ/a = me·c relations:
(7) ħ·ω = ħ·c/a = (ħ/a)·c = (me·c)·c = me·c2 = E
Of course, we note all of the formulas we have derived are interdependent. We, therefore, have no clear separation between axioms and derivations here. If anything, we are only explaining what Nature’s most fundamental laws (the Planck-Einstein relation and Einstein’s mass-energy equivalence relation) actually mean or represent. As such, all we have is a simple description of reality itself—at the smallest scale, of course! Everything that happens at larger scales involves Maxwell’s equations: that’s all electromagnetic in nature. No need for strong or weak forces, or for quarks—who invented that? Ehrenfest, Lorentz and all who suffered with truly understanding the de Broglie’s concept of the matter-wave might have been happier physicists if they would have seen these simple equations!
The gist of the matter is this: the intuition of Einstein and de Broglie in regard to the wave-nature of matter was, essentially, correct. However, de Broglie’s modeling of it as a wave packet was not: modeling matter-particles as some linear oscillation does not do the trick. It is extremely surprising no one thought of trying to think of some circular oscillation. Indeed, the interpretation of the elementary wavefunction as representing the mentioned Zitterbewegung of the electric charge solves all questions: it amounts to interpreting the real and imaginary part of the elementary wavefunction as the sine and cosine components of the orbital motion of a pointlike charge. We think that, in our 60-odd papers, we’ve shown such easy interpretation effectively does the trick of explaining all of the quantum-mechanical weirdness but, of course, it is up to our readers to judge that. 🙂
[1] See: John Stewart Bell, Speakable and unspeakable in quantum mechanics, pp. 169–172, Cambridge University Press, 1987 (quoted from Wikipedia). J.S. Bell died from a cerebral hemorrhage in 1990 – the year he was nominated for the Nobel Prize in Physics and which he, therefore, did not receive (Nobel Prizes are not awarded posthumously). He was just 62 years old then.





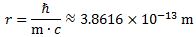
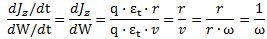

 The question which remains open, then, is the following: what is the nature of this force? In previous posts, I suggested it might be gravitational, but so here we’re back to the drawing board: we’re talking an electrical force, but applied to some mass which acquires mass because of… Well… Because of the force—because of the oscillation (the moving charge) itself. Hmm…. I need to think about this.
The question which remains open, then, is the following: what is the nature of this force? In previous posts, I suggested it might be gravitational, but so here we’re back to the drawing board: we’re talking an electrical force, but applied to some mass which acquires mass because of… Well… Because of the force—because of the oscillation (the moving charge) itself. Hmm…. I need to think about this.