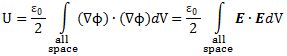Pre-scriptum (dated 26 June 2020): This post does not seem to have suffered from the attack by the dark force. However, my views on the nature of light and matter have evolved as part of my explorations of a more realist (classical) explanation of quantum mechanics. If you are reading this, then you are probably looking for not-to-difficult reading. In that case, I would suggest you read my re-write of Feynman’s introductory lecture to QM. If you want something shorter, you can also read my paper on what I believe to be the true Principles of Physics.
Original post:
I started the two previous posts attempting to justify why we need all these mathematical formulas to understand stuff: because otherwise we just keep on repeating very simplistic but nonsensical things such as ‘matter behaves (sometimes) like light’, ‘light behaves (sometimes) like matter’ or, combining both, ‘light and matter behave like wavicles’. Indeed: what does ‘like‘ mean? Like the same but different? 🙂 However, I have not said much about light so far.
Light and matter are two very different things. For matter, we have quantum mechanics. For light, we have quantum electrodynamics (QED). However, QED is not only a quantum theory about light: as Feynman pointed out in his little but exquisite 1985 book on quantum electrodynamics (QED: The Strange Theory of Light and Matter), it is first and foremost a theory about how light interacts with matter. However, let’s limit ourselves here to light.
In classical physics, light is an electromagnetic wave: it just travels on and on and on because of that wonderful interaction between electric and magnetic fields. A changing electric field induces a magnetic field, the changing magnetic field then induces an electric field, and then the changing electric field induces a magnetic field, and… Well, you got the idea: it goes on and on and on. This wonderful machinery is summarized in Maxwell’s equations – and most beautifully so in the so-called Heaviside form of these equations, which assume a charge-free vacuum space (so there are no other charges lying around exerting a force on the electromagnetic wave or the (charged) particle whom’s behavior we want to study) and they also make abstraction of other complications such as electric currents (so there are no moving charges going around either).
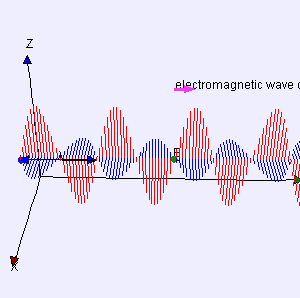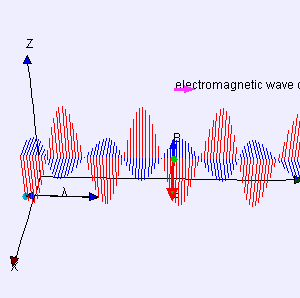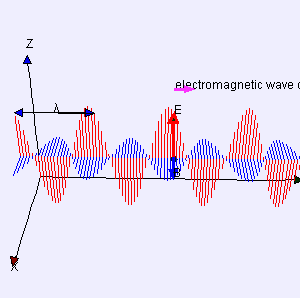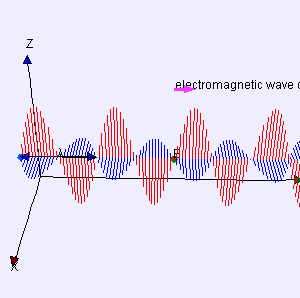
I reproduced Heaviside’s Maxwell equations below as well as an animated gif which is supposed to illustrate the dynamics explained above. [In case you wonder who’s Heaviside? Well… Check it out: he was quite a character.] The animation is not all that great but OK enough. And don’t worry if you don’t understand the equations – just note the following:
- The electric and magnetic field E and B are represented by perpendicular oscillating vectors.
- The first and third equation (∇·E = 0 and ∇·B = 0) state that there are no static or moving charges around and, hence, they do not have any impact on (the flux of) E and B.
- The second and fourth equation are the ones that are essential. Note the time derivatives (∂/∂t): E and B oscillate and perpetuate each other by inducing new circulation of B and E.
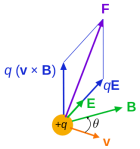
The constants μ and ε in the fourth equation are the so-called permeability (μ) and permittivity (ε) of the medium, and μ0 and ε0 are the values for these constants in a vacuum space. Now, it is interesting to note that με equals 1/c2, so a changing electric field only produces a tiny change in the circulation of the magnetic field. That’s got something to do with magnetism being a ‘relativistic’ effect but I won’t explore that here – except for noting that the final Lorentz force on a (charged) particle F = q(E + v×B) will be the same regardless of the reference frame (moving or inertial): the reference frame will determine the mixture of E and B fields, but there is only one combined force on a charged particle in the end, regardless of the reference frame (inertial or moving at whatever speed – relativistic (i.e. close to c) or not). [The forces F, E and B on a moving (charged) particle are shown below the animation of the electromagnetic wave.] In other words, Maxwell’s equations are compatible with both special as well as general relativity. In fact, Einstein observed that these equations ensure that electromagnetic waves always travel at speed c (to use his own words: “Light is always propagated in empty space with a definite velocity c which is independent of the state of motion of the emitting body.”) and it’s this observation that led him to develop his special relativity theory.
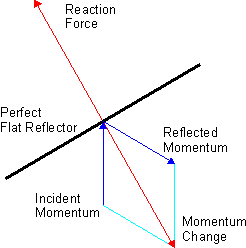
The other interesting thing to note is that there is energy in these oscillating fields and, hence, in the electromagnetic wave. Hence, if the wave hits an impenetrable barrier, such as a paper sheet, it exerts pressure on it – known as radiation pressure. [By the way, did you ever wonder why a light beam can travel through glass but not through paper? Check it out!] A very oft-quoted example is the following: if the effects of the sun’s radiation pressure on the Viking spacecraft had been ignored, the spacecraft would have missed its Mars orbit by about 15,000 kilometers. Another common example is more science fiction-oriented: the (theoretical) possibility of space ships using huge sails driven by sunlight (paper sails obviously – one should not use transparent plastic for that).
I am mentioning radiation pressure because, although it is not that difficult to explain radiation pressure using classical electromagnetism (i.e. light as waves), the explanation provided by the ‘particle model’ of light is much more straightforward and, hence, a good starting point to discuss the particle nature of light:
- Electromagnetic radiation is quantized in particles called photons. We know that because of Max Planck’s work on black body radiation, which led to Planck’s relation: E = hν. Photons are bona fide particles in the so-called Standard Model of physics: they are defined as bosons with spin 1, but zero rest mass and no electric charge (as opposed to W bosons). They are denoted by the letter or symbol γ (gamma), so that’s the same symbol that’s used to denote gamma rays. [Gamma rays are high-energy electromagnetic radiation (i.e. ‘light’) that have a very definite particle character. Indeed, because of their very short wavelength – less than 10 picometer (10×10–12 m) and high energy (hundreds of KeV – as opposed to visible light, which has a wavelength between 380 and 750 nanometer (380-750×10–9 m) and typical energy of 2 to 3 eV only (so a few hundred thousand times less), they are capable of penetrating through thick layers of concrete, and the human body – where they might damage intracellular bodies and create cancer (lead is a more efficient barrier obviously: a shield of a few centimeter of lead will stop most of them. In case you are not sure about the relation between energy and penetration depth, see the Post Scriptum.]
- Although photons are considered to have zero rest mass, they have energy and, hence, an equivalent relativistic mass (m = E/c2) and, therefore, also momentum. Indeed, energy and momentum are related through the following (relativistic) formula: E = (p2c2 + m02c4)1/2 (the non-relativistic version is simply E = p2/2m0 but – quite obviously – an approximation that cannot be used in this case – if only because the denominator would be zero). This simplifies to E = pc or p = E/c in this case. This basically says that the energy (E) and the momentum (p) of a photon are proportional, with c – the velocity of the wave – as the factor of proportionality.
- The generation of radiation pressure can then be directly related to the momentum property of photons, as shown in the diagram below – which shows how radiation force could – perhaps – propel a space sailing ship. [Nice idea, but I’d rather bet on nuclear-thermal rocket technology.]
I said in my introduction to this post that light and matter are two very different things. They are, and the logic connecting matter waves and electromagnetic radiation is not straightforward – if there is any. Let’s look at the two equations that are supposed to relate the two – the de Broglie relation and the Planck relation:
- The de Broglie relation E = hf assigns a de Broglie frequency (i.e. the frequency of a complex-valued probability amplitude function) to a particle with mass m through the mass-energy equivalence relation E = mc2. However, the concept of a matter wave is rather complicated (if you don’t think so: read the two previous posts): matter waves have little – if anything – in common with electromagnetic waves. Feynman calls electromagnetic waves ‘real’ waves (just like water waves, or sound waves, or whatever other wave) as opposed to… Well – he does stop short of calling matter waves unreal but it’s obvious they look ‘less real’ than ‘real waves’. Indeed, these complex-valued psi functions (Ψ) – for which we have to square the modulus to get the probability of something happening in space and time, or to measure the likely value of some observable property of the system – are obviously ‘something else’! [I tried to convey their ‘reality’ as well as I could in my previous post, but I am not sure I did a good job – not all really.]
- The Planck relation E = hν relates the energy of a photon – the so-called quantum of light (das Lichtquant as Einstein called it in 1905 – the term ‘photon’ was coined some 20 years later it is said) – to the frequency of the electromagnetic wave of which it is part. [That Greek symbol (ν) – it’s the letter nu (the ‘v’ in Greek is amalgamated with the ‘b’) – is quite confusing: it’s not the v for velocity.]
So, while the Planck relation (which goes back to 1905) obviously inspired Louis de Broglie (who introduced his theory on electron waves some 20 years later – in his PhD thesis of 1924 to be precise), their equations look the same but are different – and that’s probably the main reason why we keep two different symbols – f and ν – for the two frequencies.
Photons and electrons are obviously very different particles as well. Just to state the obvious:
- Photons have zero rest mass, travel at the speed of light, have no electric charge, are bosons, and so on and so on, and so they behave differently (see, for example, my post on Bose and Fermi, which explains why one cannot make proton beam lasers). [As for the boson qualification, bosons are force carriers: photons in particular mediate (or carry) the electromagnetic force.]
- Electrons do not weigh much and, hence, can attain speeds close to light (but it requires tremendous amounts of energy to accelerate them very near c) but so they do have some mass, they have electric charge (photons are electrically neutral), and they are fermions – which means they’re an entirely different ‘beast’ so to say when it comes to combining their probability amplitudes (so that’s why they’ll never get together in some kind of electron laser beam either – just like protons or neutrons – as I explain in my post on Bose and Fermi indeed).
That being said, there’s some connection of course (and that’s what’s being explored in QED):
- Accelerating electric charges cause electromagnetic radiation (so moving charges (the negatively charged electrons) cause the electromagnetic field oscillations, but it’s the (neutral) photons that carry it).
- Electrons absorb and emit photons as they gain/lose energy when going from one energy level to the other.
- Most important of all, individual photons – just like electrons – also have a probability amplitude function – so that’s a de Broglie or matter wave function if you prefer that term.
That means photons can also be described in terms of some kind of complex wave packet, just like that electron I kept analyzing in my previous posts – until I (and surely you) got tired of it. That means we’re presented with the same type of mathematics. For starters, we cannot be happy with assigning a unique frequency to our (complex-valued) de Broglie wave, because that would – once again – mean that we have no clue whatsoever where our photon actually is. So, while the shape of the wave function below might well describe the E and B of a bona fide electromagnetic wave, it cannot describe the (real or imaginary) part of the probability amplitude of the photons we would associate with that wave.
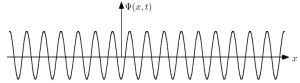 So that doesn’t work. We’re back at analyzing wave packets – and, by now, you know how complicated that can be: I am sure you don’t want me to mention Fourier transforms again! So let’s turn to Feynman once again – the greatest of all (physics) teachers – to get his take on it. Now, the surprising thing is that, in his 1985 Lectures on Quantum Electrodynamics (QED), he doesn’t really care about the amplitude of a photon to be at point x at time t. What he needs to know is:
So that doesn’t work. We’re back at analyzing wave packets – and, by now, you know how complicated that can be: I am sure you don’t want me to mention Fourier transforms again! So let’s turn to Feynman once again – the greatest of all (physics) teachers – to get his take on it. Now, the surprising thing is that, in his 1985 Lectures on Quantum Electrodynamics (QED), he doesn’t really care about the amplitude of a photon to be at point x at time t. What he needs to know is:
- The amplitude of a photon to go from point A to B, and
- The amplitude of a photon to be absorbed/emitted by an electron (a photon-electron coupling as it’s called).
And then he needs only one more thing: the amplitude of an electron to go from point A to B. That’s all he needs to explain EVERYTHING – in quantum electrodynamics that is. So that’s partial reflection, diffraction, interference… Whatever! In Feynman’s own words: “Out of these three amplitudes, we can make the whole world, aside from what goes on in nuclei, and gravitation, as always!” So let’s have a look at it.
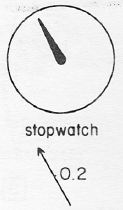
I’ve shown some of his illustrations already in the Bose and Fermi post I mentioned above. In Feynman’s analysis, photons get emitted by some source and, as soon as they do, they travel with some stopwatch, as illustrated below. The speed with which the hand of the stopwatch turns is the angular frequency of the phase of the probability amplitude, and it’s length is the modulus -which, you’ll remember, we need to square to get a probability of something, so for the illustration below we have a probability of 0.2×0.2 = 4%. Probability of what? Relax. Let’s go step by step.
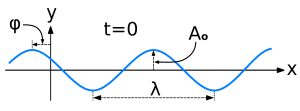
Let’s first relate this probability amplitude stopwatch to a theoretical wave packet, such as the one below – which is a nice Gaussian wave packet:
This thing really fits the bill: it’s associated with a nice Gaussian probability distribution (aka as a normal distribution, because – despite its ideal shape (from a math point of view), it actually does describe many real-life phenomena), and we can easily relate the stopwatch’s angular frequency to the angular frequency of the phase of the wave. The only thing you’ll need to remember is that its amplitude is not constant in space and time: indeed, this photon is somewhere sometime, and that means it’s no longer there when it’s gone, and also that it’s not there when it hasn’t arrived yet. 🙂 So, as you long as you remember that, Feynman’s stopwatch is a great way to represent a photon (or any particle really). [Just think of a stopwatch in your hand with no hand, but then suddenly that hand grows from zero to 0.2 (or some other random value between 0 and 1) and then shrinks back from that random value to 0 as the photon whizzes by. […] Or find some other creative interpretation if you don’t like this one. :-)]
Now, of course we do not know at what time the photon leaves the source and so the hand of the stopwatch could be at 2 o’clock, 9 o’clock or whatever: so the phase could be shifted by any value really. However, the thing to note is that the stopwatch’s hand goes around and around at a steady (angular) speed.
That’s OK. We can’t know where the photon is because we’re obviously assuming a nice standardized light source emitting polarized light with a very specific color, i.e. all photons have the same frequency (so we don’t have to worry about spin and all that). Indeed, because we’re going to add and multiply amplitudes, we have to keep it simple (the complicated things should be left to clever people – or academics). More importantly, it’s OK because we don’t need to know the exact position of the hand of the stopwatch as the photon leaves the source in order to explain phenomena like the partial reflection of light on glass. What matters there is only how much the stopwatch hand turns in the short time it takes to go from the front surface of the glass to its back surface. That difference in phase is independent from the position of the stopwatch hand as it reaches the glass: it only depends on the angular frequency (i.e. the energy of the photon, or the frequency of the light beam) and the thickness of the glass sheet. The two cases below present two possibilities: a 5% chance of reflection and a 16% chance of reflection (16% is actually a maximum, as Feynman shows in that little book, but that doesn’t matter here).
But – Hey! – I am suddenly talking amplitudes for reflection here, and the probabilities that I am calculating (by adding amplitudes, not probabilities) are also (partial) reflection probabilities. Damn ! YOU ARE SMART! It’s true. But you get the idea, and I told you already that Feynman is not interested in the probability of a photon just being here or there or wherever. He’s interested in (1) the amplitude of it going from the source (i.e. some point A) to the glass surface (i.e. some other point B), and then (2) the amplitude of photon-electron couplings – which determine the above amplitudes for being reflected (i.e. being (back)scattered by an electron actually).
So what? Well… Nothing. That’s it. I just wanted you to give some sense of de Broglie waves for photons. The thing to note is that they’re like de Broglie waves for electrons. So they are as real or unreal as these electron waves, and they have close to nothing to do with the electromagnetic wave of which they are part. The only thing that relates them with that real wave so to say, is their energy level, and so that determines their de Broglie wavelength. So, it’s strange to say, but we have two frequencies for a photon: E= hν and E = hf. The first one is the Planck relation (E= hν): it associates the energy of a photon with the frequency of the real-life electromagnetic wave. The second is the de Broglie relation (E = hf): once we’ve calculated the energy of a photon using E= hν, we associate a de Broglie wavelength with the photon. So we imagine it as a traveling stopwatch with angular frequency ω = 2πf.
So that’s it (for now). End of story.
[…]
Now, you may want to know something more about these other amplitudes (that’s what I would want), i.e. the amplitude of a photon to go from A to B and this coupling amplitude and whatever else that may or may not be relevant. Right you are: it’s fascinating stuff. For example, you may or may not be surprised that photons have an amplitude to travel faster or slower than light from A to B, and that they actually have many amplitudes to go from A to B: one for each possible path. [Does that mean that the path does not have to be straight? Yep. Light can take strange paths – and it’s the interplay (i.e. the interference) between all these amplitudes that determines the most probable path – which, fortunately (otherwise our amplitude theory would be worthless), turns out to be the straight line.] We can summarize this in a really short and nice formula for the P(A to B) amplitude [note that the ‘P’ stands for photon, not for probability – Feynman uses an E for the related amplitude for an electron, so he writes E(A to B)].
However, I won’t make this any more complicated right now and so I’ll just reveal that P(A to B) depends on the so-called spacetime interval. This spacetime interval (I) is equal to I = (z2– z1)2 + (y2– y1)2 + (x2– x1)2 – (t2– t1)2, with the time and spatial distance being measured in equivalent units (so we’d use light-seconds for the unit of distance or, for the unit of time, the time it takes for light to travel one meter). I am sure you’ve heard about this interval. It’s used to explain the famous light cone – which determines what’s past and future in respect to the here and now in spacetime (or the past and present of some event in spacetime) in terms of
- What could possibly have impacted the here and now (taking into account nothing can travel faster than light – even if we’ve mentioned some exceptions to this already, such as the phase velocity of a matter wave – but so that’s not a ‘signal’ and, hence, not in contradiction with relativity)?
- What could possible be impacted by the here and now (again taking into account that nothing can travel faster than c)?
In short, the light cone defines the past, the here, and the future in spacetime in terms of (potential) causal relations. However, as this post has – once again – become too long already, I’ll need to write another post to discuss these other types of amplitudes – and how they are used in quantum electrodynamics. So my next post should probably say something about light-matter interaction, or on photons as the carriers of the electromagnetic force (both in light as well as in an atom – as it’s the electromagnetic force that keeps an electron in orbit around the (positively charged) nucleus). In case you wonder, yes, that’s Feynman diagrams – among other things.
Post scriptum: On frequency, wavelength and energy – and the particle- versus wave-like nature of electromagnetic waves
I wrote that gamma waves have a very definite particle character because of their very short wavelength. Indeed, most discussions of the electromagnetic spectrum will start by pointing out that higher frequencies or shorter wavelengths – higher frequency (f) implies shorter wavelength (λ) because the wavelength is the speed of the wave (c in this case) over the frequency: λ = c/f – will make the (electromagnetic) wave more particle-like. For example, I copied two illustrations from Feynman’s very first Lectures (Volume I, Lectures 2 and 5) in which he makes the point by showing
- The familiar table of the electromagnetic spectrum (we could easily add a column for the wavelength (just calculate λ = c/f) and the energy (E = hf) besides the frequency), and
- An illustration that shows how matter (a block of carbon of 1 cm thick in this case) looks like for an electromagnetic wave racing towards it. It does not look like Gruyère cheese, because Gruyère cheese is cheese with holes: matter is huge holes with just a tiny little bit of cheese ! Indeed, at the micro-level, matter looks like a lot of nothing with only a few tiny specks of matter sprinkled about!
And so then he goes on to describe how ‘hard’ rays (i.e. rays with short wavelengths) just plow right through and so on and so on.
Now it will probably sound very stupid to non-autodidacts but, for a very long time, I was vaguely intrigued that the amplitude of a wave doesn’t seem to matter when looking at the particle- versus wave-like character of electromagnetic waves. Electromagnetic waves are transverse waves so they oscillate up and down, perpendicular to the direction of travel (as opposed to longitudinal waves, such as sound waves or pressure waves for example: these oscillate back and forth – in the same direction of travel). And photon paths are represented by wiggly lines, so… Well, you may not believe it but that’s why I stupidly thought it’s the amplitude that should matter, not the wavelength.
Indeed, the illustration below – which could be an example of how E or B oscillates in space and time – would suggest that lower amplitudes (smaller A’s) are the key to ‘avoiding’ those specks of matter. And if one can’t do anything about amplitude, then one may be forgiven to think that longer wavelengths – not shorter ones – are the key to avoiding those little ‘obstacles’ presented by atoms or nuclei in some crystal or non-crystalline structure. [Just jot it down: more wiggly lines increase the chance of hitting something.] But… Both lower amplitudes as well as longer wavelengths imply less energy. Indeed, the energy of a wave is, in general, proportional to the square of its amplitude and electromagnetic waves are no exception in this regard. As for wavelength, we have Planck’s relation. So what’s wrong in my very childish reasoning?
As usual, the answer is easy for those who already know it: neither wavelength nor amplitude have anything to do with how much space this wave actually takes as it propagates. But of course! You didn’t know that? Well… Sorry. Now I do. The vertical y axis might measure E and B indeed, but the graph and the nice animation above should not make you think that these field vectors actually occupy some space. So you can think of electromagnetic waves as particle waves indeed: we’ve got ‘something’ that’s traveling in a straight line, and it’s traveling at the speed of light. That ‘something’ is a photon, and it can have high or low energy. If it’s low-energy, it’s like a speck of dust: even if it travels at the speed of light, it is easy to deflect (i.e. scatter), and the ’empty space’ in matter (which is, of course, not empty but full of all kinds of electromagnetic disturbances) may well feel like jelly to it: it will get stuck (read: it will be absorbed somewhere or not even get through the first layer of atoms at all). If it’s high-energy, then it’s a different story: then the photon is like a tiny but very powerful bullet – same size as the speck of dust, and same speed, but much and much heavier. Such ‘bullet’ (e.g. a gamma ray photon) will indeed have a tendency to plow through matter like it’s air: it won’t care about all these low-energy fields in it.
It is, most probably, a very trivial point to make, but I thought it’s worth doing so.
[When thinking about the above, also remember the trivial relationship between energy and momentum for photons: p = E/c, so more energy means more momentum: a heavy truck crashing into your house will create more damage than a Mini at the same speed because the truck has much more momentum. So just use the mass-energy equivalence (E = mc2) and think about high-energy photons as armored vehicles and low-energy photons as mom-and-pop cars.]
Some content on this page was disabled on June 16, 2020 as a result of a DMCA takedown notice from The California Institute of Technology. You can learn more about the DMCA here:
https://wordpress.com/support/copyright-and-the-dmca/
Some content on this page was disabled on June 20, 2020 as a result of a DMCA takedown notice from Michael A. Gottlieb, Rudolf Pfeiffer, and The California Institute of Technology. You can learn more about the DMCA here:https://wordpress.com/support/copyright-and-the-dmca/
Some content on this page was disabled on June 20, 2020 as a result of a DMCA takedown notice from Michael A. Gottlieb, Rudolf Pfeiffer, and The California Institute of Technology. You can learn more about the DMCA here:

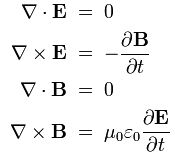




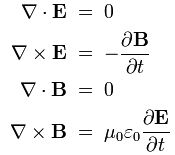
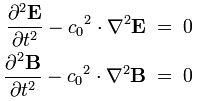
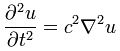
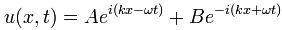
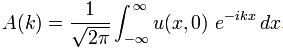
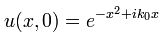
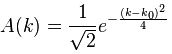
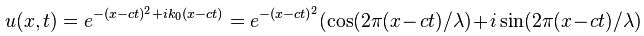
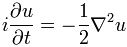

 Now if we plug that in the equation above, we get our time-dependent Schrödinger equation:
Now if we plug that in the equation above, we get our time-dependent Schrödinger equation: