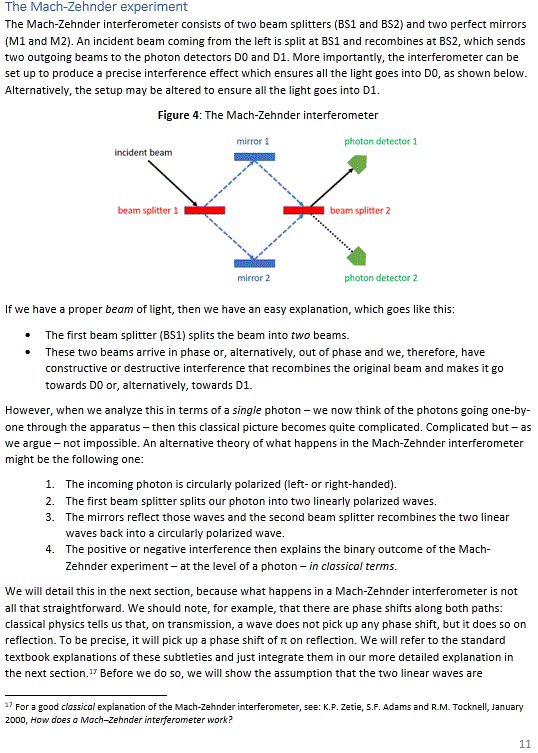
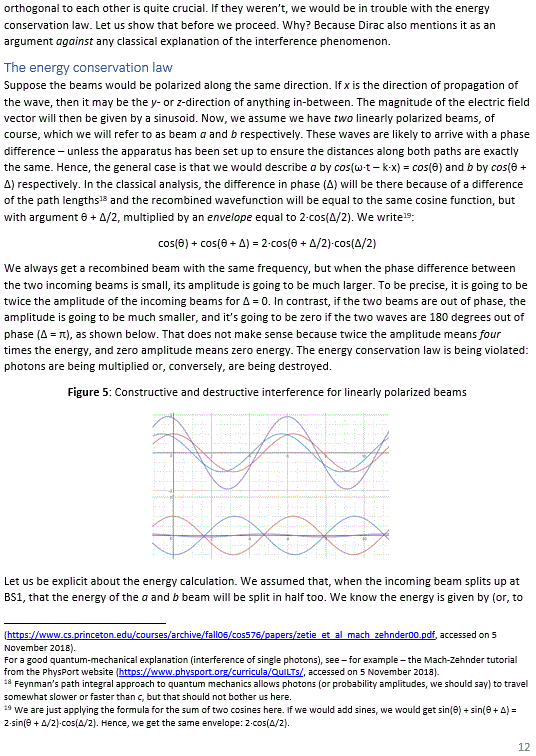
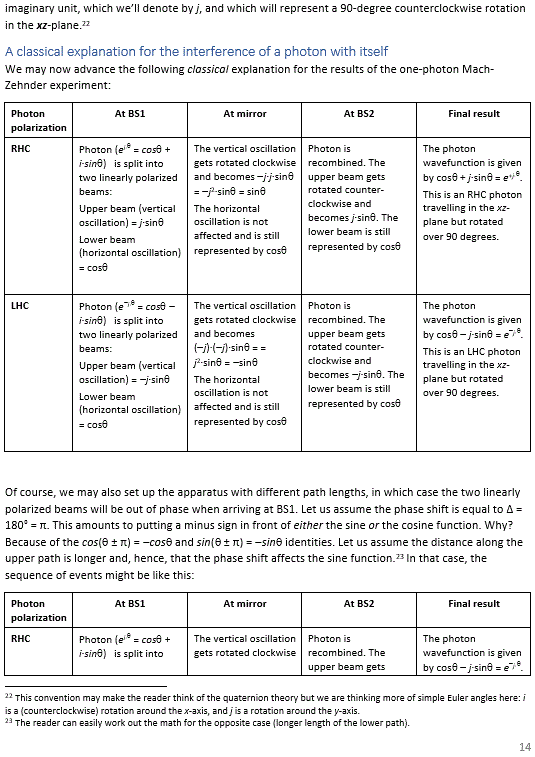
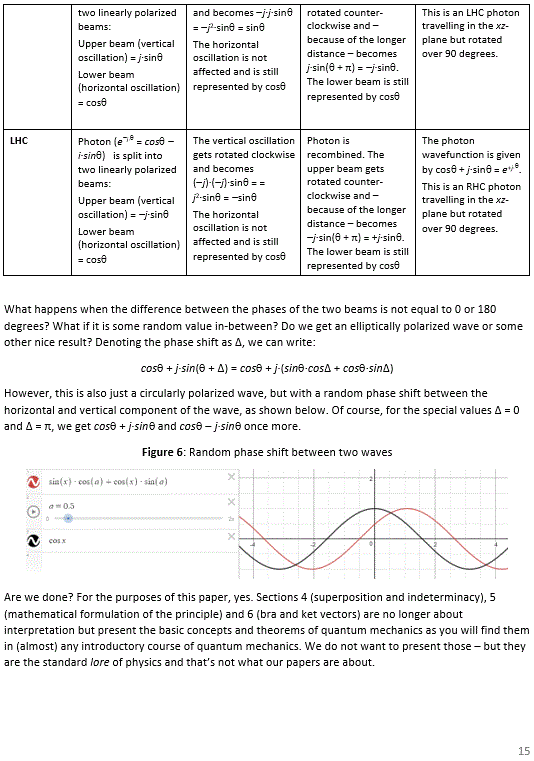
Pre-scriptum (dated 27 June 2020): Two illustrations in this post were deleted by the dark force. We will not substitute them. The reference is given and it will help you to look them up yourself. In fact, we think it will greatly advance your understanding if you do so. Mr. Gottlieb may actually have done us a favor by trying to pester us.
Electrons, atoms, elementary particles and wave equations
The New Zealander Ernest Rutherford came to be known as the father of nuclear physics. He was the first to provide a reliable estimate of the order of magnitude of the size of the nucleus. To be precise, in the 1921 paper which we will discuss here, he came up with an estimate of about 15 fm for massive nuclei, which is the current estimate for the size of an uranium nucleus. His experiments also helped to significantly enhance the Bohr model of an atom, culminating – just before WW I started – in the Bohr-Rutherford model of an atom (E. Rutherford, Phil. Mag. 27, 488).
The Bohr-Rutherford model of an atom explained the (gross structure of the) hydrogen spectrum perfectly well, but it could not explain its finer structure—read: the orbital sub-shells which, as we all know now (but not very well then), result from the different states of angular momentum of an electron and the associated magnetic moment.
The issue is probably best illustrated by the two diagrams below, which I copied from Feynman’s Lectures. As you can see, the idea of subshells is not very relevant when looking at the gross structure of the hydrogen spectrum because the energy levels of all subshells are (very nearly) the same. However, the Bohr model of an atom—which is nothing but an exceedingly simple application of the E = h·f equation (see p. 4-6 of my paper on classical quantum physics)—cannot explain the splitting of lines for a lithium atom, which is shown in the diagram on the right. Nor can it explain the splitting of spectral lines when we apply a stronger or weaker magnetic field while exciting the atoms so as to induce emission of electromagnetic radiation.
Schrödinger’s wave equation solves that problem—which is why Feynman and other modern physicists claim this equation is “the most dramatic success in the history of the quantum mechanics” or, more modestly, a “key result in quantum mechanics” at least!
Such dramatic statements are exaggerated. First, an even finer analysis of the emission spectrum (of hydrogen or whatever other atom) reveals that Schrödinger’s wave equation is also incomplete: the hyperfine splitting, the Zeeman splitting (anomalous or not) or the (in)famous Lamb shift are to be explained not only in terms of the magnetic moment of the electron but also in terms of the magnetic moment of the nucleus and its constituents (protons and neutrons)—or of the coupling between those magnetic moments (we may refer to our paper on the Lamb shift here). This cannot be captured in a wave equation: second-order differential equations are – quite simply – not sophisticated enough to capture the complexity of the atomic system here.
Also, as we pointed out previously, the current convention in regard to the use of the imaginary unit (i) in the wavefunction does not capture the spin direction and, therefore, makes abstraction of the direction of the magnetic moment too! The wavefunction therefore models theoretical spin-zero particles, which do not exist. In short, we cannot hope to represent anything real with wave equations and wavefunctions.
More importantly, I would dare to ask this: what use is an ‘explanation’ in terms of a wave equation if we cannot explain what the wave equation actually represents? As Feynman famously writes: “Where did we get it from? Nowhere. It’s not possible to derive it from anything you know. It came out of the mind of Schrödinger, invented in his struggle to find an understanding of the experimental observations of the real world.” Our best guess is that it, somehow, models (the local diffusion of) energy or mass densities as well as non-spherical orbital geometries. We explored such interpretations in our very first paper(s) on quantum mechanics, but the truth is this: we do not think wave equations are suitable mathematical tools to describe simple or complex systems that have some internal structure—atoms (think of Schrödinger’s wave equation here), electrons (think of Dirac’s wave equation), or protons (which is what some others tried to do, but I will let you do some googling here yourself).
We need to get back to the matter at hand here, which is Rutherford’s idea of an electron back in 1921. What can we say about it?
Rutherford’s contributions to the 1921 Solvay Conference
From what you know, and from what I write above, you will understand that Rutherford’s research focus was not on electrons: his prime interest was in explaining the atomic structure and in solving the mysteries of nuclear radiation—most notably the emission of alpha– and beta-particles as well as highly energetic gamma-rays by unstable or radioactive nuclei. In short, the nature of the electron was not his prime interest. However, this intellectual giant was, of course, very much interested in whatever experiment or whatever theory that might contribute to his thinking, and that explains why, in his contribution to the 1921 Solvay Conference—which materialized as an update of his seminal 1914 paper on The Structure of the Atom—he devotes considerable attention to Arthur Compton’s work on the scattering of light from electrons which, at the time (1921), had not even been published yet (Compton’s seminal article on (Compton) scattering was published in 1923 only).
It is also very interesting that, in the very same 1921 paper—whose 30 pages are more than a multiple of his 1914 article and later revisions of it (see, for example, the 1920 version of it, which actually has wider circulation on the Internet)—Rutherford also offers some short reflections on the magnetic properties of electrons while referring to Parson’s ring current model which, in French, he refers to as “l’électron annulaire de Parson.” Again, it is very strange that we should translate Rutherford’s 1921 remarks back in English—as we are sure the original paper must have been translated from English to French rather than the other way around.
However, it is what it is, and so here we do what we have to do: we give you a free translation of Rutherford’s remarks during the 1921 Solvay Conference on the state of research regarding the electron at that time. The reader should note these remarks are buried in a larger piece on the emission of β particles by radioactive nuclei which, as it turns out, are nothing but high-energy electrons (or their anti-matter counterpart—positrons). In fact, we should—before we proceed—draw attention to the fact that the physicists at the time had no clear notion of the concepts of protons and neutrons.
This is, indeed, another remarkable historical contribution of the 1921 Solvay Conference because, as far as I know, this is the first time Rutherford talks about the neutron hypothesis. It is quite remarkable he does not advance the neutron hypothesis to explain the atomic mass of atoms combining what we know think of as protons and neutrons (Rutherford regularly talks of a mix of ‘positive and negative electrons’ in the nucleus—neither the term proton or neutron was in use at the time) but as part of a possible explanation of nuclear fusion reactions in stars or stellar nebulae. This is, indeed, his response to a question during the discussions on Rutherford’s paper on the possibility of nuclear synthesis in stars or nebulae from the French physicist Jean Baptise Perrin who, independently from the American chemist William Draper Harkins, had proposed the possibility of hydrogen fusion just the year before (1919):
“We can, in fact, think of enormous energies being released from hydrogen nuclei merging to form helium—much larger energies than what can come from the Kelvin-Helmholtz mechanism. I have been thinking that the hydrogen in the nebulae might come from particles which we may refer to as ‘neutrons’: these would consist of a positive nucleus with an electron at an exceedingly small distance (“un noyau positif avec un électron à toute petite distance”). These would mediate the assembly of the nuclei of more massive elements. It is, otherwise, difficult to understand how the positively charged particles could come together against the repulsive force that pushes them apart—unless we would envisage they are driven by enormous velocities.”
We may add that, just to make sure he get this right, Rutherford is immediately requested to elaborate his point by the Danish physicist Martin Knudsen: “What’s the difference between a hydrogen atom and this neutron?”—which Rutherford simply answers as follows: “In a neutron, the electron would be very much closer to the nucleus.” In light of the fact that it was only in 1932 that James Chadwick would experimentally prove the existence of neutrons (and positively charged protons), we are, once again, deeply impressed by the the foresight of Rutherford and the other pioneers here: the predictive power of their theories and ideas is, effectively, truly amazing by any standard—including today’s. I should, perhaps, also add that I fully subscribe to Rutherford’s intuition that a neutron should be a composite particle consisting of a proton and an electron—but that’s a different discussion altogether.
We must come back to the topic of this post, which we will do now. Before we proceed, however, we should highlight one other contextual piece of information here: at the time, very little was known about the nature of α and β particles. We now know that beta-particles are electrons, and that alpha-particles combine two protons and two neutrons. That was not known in the 1920s, however: Rutherford and his associates could basically only see positive or negative particles coming out of these radioactive processes. This further underscores how much knowledge they were able to gain from rather limited sets of data.
Rutherford’s idea of an electron in 1921
So here is the translation of some crucial text. Needless to say, the italics, boldface and additions between [brackets] are not Rutherford’s but mine, of course.
“We may think the same laws should apply in regard to the scattering [“diffusion”] of α and β particles. [Note: Rutherford noted, earlier in his paper, that, based on the scattering patterns and other evidence, the force around the nucleus must respect the inverse square law near the nucleus—moreover, it must also do so very near to it.] However, we see marked differences. Anyone who has carefully studied the trajectories [photographs from the Wilson cloud chamber] of beta-particles will note the trajectories show a regular curvature. Such curved trajectories are even more obvious when they are illuminated by X-rays. Indeed, A.H. Compton noted that these trajectories seem to end in a converging helical path turning right or left. To explain this, Compton assumes the electron acts like a magnetic dipole whose axis is more or less fixed, and that the curvature of its path is caused by the magnetic field [from the (paramagnetic) materials that are used].
Further examination would be needed to make sure this curvature is not some coincidence, but the general impression is that the hypothesis may be quite right. We also see similar curvature and helicity with α particles in the last millimeters of their trajectories. [Note: α-particles are, obviously, also charged particles but we think Rutherford’s remark in regard to α particles also following a curved or helical path must be exaggerated: the order of magnitude of the magnetic moment of protons and neutrons is much smaller and, in any case, they tend to cancel each other out. Also, because of the rather enormous mass of α particles (read: helium nuclei) as compared to electrons, the effect would probably not be visible in a Wilson cloud chamber.]
The idea that an electron has magnetic properties is still sketchy and we would need new and more conclusive experiments before accepting it as a scientific fact. However, it would surely be natural to assume its magnetic properties would result from a rotation of the electron. Parson’s ring electron model [“électron annulaire“] was specifically imagined to incorporate such magnetic polarity [“polarité magnétique“].
A very interesting question here would be to wonder whether such rotation would be some intrinsic property of the electron or if it would just result from the rotation of the electron in its atomic orbital around the nucleus. Indeed, James Jeans usefully reminded me any asymmetry in an electron should result in it rotating around its own axis at the same frequency of its orbital rotation. [Note: The reader can easily imagine this: think of an asymmetric object going around in a circle and returning to its original position. In order to return to the same orientation, it must rotate around its own axis one time too!]
We should also wonder if an electron might acquire some rotational motion from being accelerated in an electric field and if such rotation, once acquired, would persist when decelerating in an(other) electric field or when passing through matter. If so, some of the properties of electrons would, to some extent, depend on their past.”
Each and every sentence in these very brief remarks is wonderfully consistent with modern-day modelling of electron behavior. We should add, of course, non-mainstream modeling of electrons but the addition is superfluous because mainstream physicists stubbornly continue to pretend electrons have no internal structure, and nor would they have any physical dimension. In light of the numerous experimental measurements of the effective charge radius as well as of the dimensions of the physical space in which photons effectively interfere with electrons, such mainstream assumptions seem completely ridiculous. However, such is the sad state of physics today.
Thinking backward and forward
We think that it is pretty obvious that Rutherford and others would have been able to adapt their model of an atom to better incorporate the magnetic properties not only of electrons but also of the nucleus and its constituents (protons and neutrons). Unfortunately, scientists at the time seem to have been swept away by the charisma of Bohr, Heisenberg and others, as well as by the mathematical brilliance of the likes of Sommerfeld, Dirac, and Pauli.
The road then was taken then has not led us very far. We concur with Oliver Consa’s scathing but essentially correct appraisal of the current sorry state of physics:
“QED should be the quantized version of Maxwell’s laws, but it is not that at all. QED is a simple addition to quantum mechanics that attempts to justify two experimental discrepancies in the Dirac equation: the Lamb shift and the anomalous magnetic moment of the electron. The reality is that QED is a bunch of fudge factors, numerology, ignored infinities, hocus-pocus, manipulated calculations, illegitimate mathematics, incomprehensible theories, hidden data, biased experiments, miscalculations, suspicious coincidences, lies, arbitrary substitutions of infinite values and budgets of 600 million dollars to continue the game. Maybe it is time to consider alternative proposals. Winter is coming.”
I would suggest we just go back where we went wrong: it may be warmer there, and thinking both backward as well as forward must, in any case, be a much more powerful problem solving technique than relying only on expert guessing on what linear differential equation(s) might give us some S-matrix linking all likely or possible initial and final states of some system or process. 🙂
Post scriptum: The sad state of physics is, of course, not limited to quantum electrodynamics only. We were briefly in touch with the PRad experimenters who put an end to the rather ridiculous ‘proton radius puzzle’ by re-confirming the previously established 0.83-0.84 range for the effective charge radius of a proton: we sent them our own classical back-of-the-envelope calculation of the Compton scattering radius of a proton based on the ring current model (see p. 15-16 of our paper on classical physics), which is in agreement with these measurements and courteously asked what alternative theories they were suggesting. Their spokesman replied equally courteously:
“There is no any theoretical prediction in QCD. Lattice [theorists] are trying to come up [with something] but that will take another decade before any reasonable number [may come] from them.”
This e-mail exchange goes back to early February 2020. There has been no news since. One wonders if there is actually any real interest in solving puzzles. The physicist who wrote the above may have been nominated for a Nobel Prize in Physics—I surely hope so because, in contrast to some others, he and his team surely deserve one— but I think it is rather incongruous to finally firmly establish the size of a proton while, at the same time, admit that protons should not have any size according to mainstream theory—and we are talking the respected QCD sector of the equally respected Standard Model here!
We understand, of course! As Freddy Mercury famously sang: The Show Must Go On.