Note: I have published a paper that is very coherent and fully explains this so-called God-given number. There is nothing magical about it. It is just a scaling constant. Check it out: The Meaning of the Fine-Structure Constant. No ambiguity. No hocus-pocus.
Jean Louis Van Belle, 23 December 2018
Original post:
In my previous post, I explained why the fine-structure constant α is not a ‘magical’ number, even if it relates all fundamental properties of the electron: its mass, its energy, its charge, its radius, its photon scattering cross-section (i.e. the Bohr radius, or the size of the atom really) and, finally, the coupling constant for photon-electron interactions. The key to such understanding of α was the model of an electron as a tiny ball of charge. As such, we have two energy formulas for it. One is the energy that’s needed to assemble the charge from infinitely dispersed infinitesimal charges, which we denoted as Uelec. The other formula is the energy of the field of the tiny ball of charge, which we denoted as Eelec.
The formula for Eelec is calculated using the formula for the field momentum of a moving charge and, using the m = E/c2 mas-energy equivalence relationship, is equivalent to the electromagnetic mass. We went through the derivation in our previous post, so let me just jot down the result:
The second formula depends on what ball of charge we’re thinking of, because the formulas for a charged sphere and a spherical shell of charge are different: both have the same structure as the relationship above (so the energy is also proportional to the square of the electron charge and inversely proportional to the radius a), but the constant of proportionality is different. For a sphere of charge, we write:
For a spherical shell of charge we write:

To compare the formulas, you need to note that the square of the electron charge e in the formula for the field energy is equal to e2 = qe2/4πε0 = ke·qe2. So we multiply the square of the actual electron charge by the Coulomb constant ke = 1/4πε0. As you can see, the three formulas have exactly the same form then. It’s just the proportionality constant that’s different: it’s 2/3, 3/5 and 1/2 respectively. It’s interesting to quickly reflect on the dimensions here: [ke] ≈ 9×109 N·m2/C2, so e2 is expressed in N·m2. That makes the units come out alright, as we divide by a (so that’s in meter) and so we get the energy in joule (which is newton·meter). In fact, now that we’re here, let’s quickly calculate the value of e2: it’s that ke·qe2 product, so it’s equal to 2.3×10−28 N·m2. We can quickly check this value because we know that the classical electron radius is equal to:
So we divide 2.3×10−28 N·m2 by mec2 ≈ 8.2×10−14 J, so we get r0 ≈ 2.82×10−15 m. So we’re spot on! Why did I do this check? Not really to check what I wrote. It’s more to show what’s going on. We’ve got yet another formula relating the energy and the radius of an electron here, so now we have three. In fact we have more because the formula for Uelec depends on the finer details of our model for the electron (sphere versus shell, uniform versus non-uniform distribution):
- Eelec = (2/3)·(e2/a): This is the formula for the energy of the field, so we may all it is external energy.
- Uelec = (3/5)·(e2/a), or Uelec = (1/2)·(e2/a): This is the energy needed to assemble our electron, so we might, perhaps, call it its internal energy. The first formula assumes our electron is a uniformly charged sphere. The second assumes all charges sit on the surface of the sphere. If we drop the assumption of the charge having to be uniformly distributed, we’ll find yet another formula.
- mec2 = e2/r0: This is the energy associated with the so-called classical electron radius (r0) and the electron’s rest mass (me).
In our previous posts, we assumed the last equation was the right one. Why? Because it’s the one that’s been verified experimentally. The discrepancies between the various proportionality coefficients – i.e. the difference between 2/3 and 1, basically – are to be explained because of the binding forces within the electron, without which the electron would just ‘explode’, as the French physicist and polymath Henri Poincaré famously put it. Indeed, if the electron is a little ball of negative charge, the repulsive forces between its parts should rip it apart. So we will not say anything more about this. You can have fun yourself by googling all the various theories that try to model these binding forces. [I may do the same some day, but now I’ve got other priorities: I want to move to Feynman’s third volume of Lectures, which is devoted to quantum physics only, so I look very much forward to that.]
In this post, I just wanted to reflect once more on what constants are really fundamental and what constants are somewhat less fundamental. From all what I wrote in my previous post, I said there were three:
- The fine-structure constant α, which is a dimensionless number.
- Planck’s constant h, whose dimension is joule·second, so that’s the dimension of action.
- The speed of light c, whose dimension is that of a velocity.
The three are related through the following expression:
This is an interesting expression. Let’s first check its dimension. We already explained that e2 is expressed in N·m2. That’s rather strange, because it means the dimension of e itself is N1/2·m: what’s the square root of a force of one newton? In fact, to interpret the formula above, it’s probably better to re-write e2 as e2 = qe2/4πε0 = ke·qe2. That shows you how the electron charge and Coulomb’s constant are related. Of course, they are part and parcel of one and the same force law: Coulomb’s law. We don’t need anything else, except for relativity theory, because we need to explain the magnetic force as well—and that we can do because magnetism is just a relativistic effect. Think of the field momentum indeed: the magnetic field comes into play only when we start to move our electron. The relativity effect is captured by c in that formula for α above. As for ħ, ħ = h/2π comes with the E = h·f equation, which links us to the electron’s Compton wavelength λ through the de Broglie relation λ = h/p.
The point is: we should probably not look at α as a ‘fundamental physical constant’. It’s e2 that’s the third fundamental constant, besides h and c. Indeed, it’s from e2 that all the rest follows: the electron’s internal energy, its external energy, and its radius, and then all the rest by combining stuff with other stuff.
Now, we took the magic out of α by doing what we did in the previous posts, and that’s to combine stuff with other stuff, and so now you may think I am putting the magic back in with that formula for α, which seems to define α in terms of the three mentioned ‘fundamental’ constants. That’s not the case: this relation comes out of all of the other relationships we found, and so it’s nothing new really. It’s actually not a definition of α: it just does what it does, and that’s to relate α to the ‘fundamental’ physical constants behind.
So… No new magic. In fact, I want to close this post by taking away even more of the magic. If you read my previous post, I said that α was ‘God’s cut-off factor’ 🙂 ensuring our energy functions do not blow up, but I also said it was impossible to say why he chose 0.00729735256 as the cut-off factor. The question is actually easily answered by thinking about those two formulas we had for the internal and external energy respectively. Let’s re-write them in natural units and, temporarily, two different subscripts for α, so we write:
- Eelec = αe/r0: This is the formula for the energy of the field.
- Uelec = αu/r0: This is the energy needed to assemble our electron.
Both energies are determined by the above-mentioned laws, i.e. Coulomb’s Law and the theory of relativity, so α has got nothing to do what that. However, both energies have to be the same, and so αe has to be equal to αu. In that sense, α is, quite simply, a proportionality constant that achieves that equality. Now that explains why we can derive α from the three other constants which, as mentioned above, are probably more fundamental. In fact, we’ve got only three degrees of freedom here, so if we chose c, h and e as ‘fundamental’, then α isn’t any more.
The underlying deep question behind it all is why those two energies should be equal. Why would our electron have some internal energy if it’s elementary? The answer to that question is: because it has some non-zero radius, and it has some non-zero radius because we don’t want our formula for the field energy (or the field momentum) to blow up. Now, if it has some radius, then it has to have some internal energy.
You’ll say: that makes sense, but it doesn’t answer the question. Why would it have internal energy, with or without a zero radius? If an electron is an elementary particle, then it’s really elementary, isn’t? And so then we shouldn’t try to ‘assemble’ it from an infinite number of infinitesimally small charges. You’re right, and here we can also note that the fact that the electron doesn’t blow up is firm evidence it’s very elementary indeed.
I should also note that Feynman actually doesn’t talk about the energy that’s needed to assemble a charge: he gets his Uelec = (1/2)·(e2/a) by calculating the external field energy for a spherical shell of charge, and he sticks to it—presumably because it’s the same field for a uniform or non-uniform sphere of charge. He only notes there has to be some radius because, if not, the formula he uses blows up, indeed. So – who knows? – perhaps he doesn’t quite believe that formula for the internal energy is relevant either.
So perhaps there is no internal energy indeed. Perhaps there’s just the energy of the field. So… Well… I can’t say much about this… Except… Well… Perhaps just one more thing. Let me note something that, I hope, you noticed as well: the ke·qe2 is the numerator in Coulomb’s Law itself. You also know that energy equals force times distance. So if we divide both sides by r0, we get Coulomb’s Law itself Felec = ke·qe2/r02. The only thing is: what’s the distance? It’s one charge only, and there is no distance between one charge, is there? Well… Yes and no. I have been thinking that the requirement of the internal and external energies being equal resembles the statement that the forces between two charges are equal and opposite. That ties in with the idea of the internal energy itself: remember we were basically talking forces between infinitesimally small elements of charge within the electron itself? So r0 is, perhaps, some average distance or so. There must be some way of thinking of it like that. But… Well… Which one exactly?
This kind of reflection may not make sense. Who knows? I obviously need to think all of this through and so this post is, indeed, just a bunch of reflections for which I will have more time later—hopefully. 🙂 Perhaps we’re all just pushing the matter too far. Perhaps we should just accept that the external energy has that 2/3 factor but that the actual energy of the electron should also include the equivalent energy of some binding force that holds the electron together. Well… In any case. That’s all I am going to do on this extremely complicated matter. It’s time to move indeed! So the point to take home here is probably just this:
- When calculating the radius of an electron using classical theory, we get in trouble: not only do we find different radii, but the radii that we find do not respect the E = mec2 law. It’s only the mec2 = e2/r0 that’s relativistically correct.
- That suggests the electron also has some non-electric mass, which are referred to as ‘binding forces’ or ‘Poincaré stresses’, but which remain to be explained convincingly.
- All of this shouldn’t surprise us: for all we know, the electron is something fuzzy. 🙂
So my next posts will focus on the ‘essentials’ preparing for Feynman’s Volume on quantum mechanics. Those ‘essentials’ will still involve some classical stuff but, as you will see, even more contradictions, that – hopefully! – will then be solved in the quantum-mechanical picture of it all. 🙂
Some content on this page was disabled on June 16, 2020 as a result of a DMCA takedown notice from The California Institute of Technology. You can learn more about the DMCA here:
https://wordpress.com/support/copyright-and-the-dmca/
Some content on this page was disabled on June 20, 2020 as a result of a DMCA takedown notice from Michael A. Gottlieb, Rudolf Pfeiffer, and The California Institute of Technology. You can learn more about the DMCA here:https://wordpress.com/support/copyright-and-the-dmca/
Some content on this page was disabled on June 20, 2020 as a result of a DMCA takedown notice from Michael A. Gottlieb, Rudolf Pfeiffer, and The California Institute of Technology. You can learn more about the DMCA here:

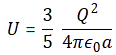

2 thoughts on “Taking the magic out of God’s number: some additional reflections”