Pre-scriptum (dated 26 June 2020): This post did not suffer from the DMCA take-down of some material. It is, therefore, still quite readable—even if my views on these matters have evolved quite a bit as part of my realist interpretation of QM. However, I now think de Broglie’s intuition in regard to particles being waves was correct but that he should have used a circular rather than a linear wave concept. Also, the idea of a particle being some wave packet is erroneous. It leads to the kind of contradictions I already start mentioning here, such as super-luminous velocities and other nonsense. Such critique is summarized in my paper on de Broglie’s wave concept. I also discuss it in the context of analyzing wavefunction math in the context of signal transmission in a crystal lattice.
Original post:
In my previous posts, I introduced a lot of wave formulas. They are essential to understanding waves – both real ones (e.g. electromagnetic waves) as well as probability amplitude functions. Probability amplitude function is quite a mouthful so let me call it a matter wave, or a de Broglie wave. The formulas are necessary to create true understanding – whatever that means to you – because otherwise we just keep on repeating very simplistic but nonsensical things such as ‘matter behaves (sometimes) like light’, ‘light behaves (sometimes) like matter’ or, combining both, ‘light and matter behave like wavicles’. Indeed: what does ‘like‘ mean? Like the same but different? 🙂 So that means it’s different. Let’s therefore re-visit the matter wave (i.e. the de Broglie wave) and point out the differences with light waves.
In fact, this post actually has its origin in a mistake in a post scriptum of a previous post (An Easy Piece: On Quantum Mechanics and the Wave Function), in which I wondered what formula to use for the energy E in the (first) de Broglie relation E = hf (with f the frequency of the matter wave and h the Planck constant). Should we use (a) the kinetic energy of the particle, (b) the rest mass (mass is energy, remember?), or (c) its total energy? So let us first re-visit these de Broglie relations which, you’ll remember, relate energy and momentum to frequency (f) and wavelength (λ) respectively with the Planck constant as the factor of proportionality:
E = hf and p = h/λ
The de Broglie wave
I first tried kinetic energy in that E = hf equation. However, if you use the kinetic energy formula (K.E. = mv2/2, with v the velocity of the particle), then the second de Broglie relation (p = h/λ) does not come out right. The second de Broglie relation has the wavelength λ on the right side, not the frequency f. But it’s easy to go from one to the other: frequency and wavelength are related through the velocity of the wave (v). Indeed, the number of cycles per second (i.e. the frequency f) times the length of one cycle (i.e. the wavelength λ) gives the distance traveled by the wave per second, i.e. its velocity v. So fλ = v. Hence, using that kinetic energy formula and that very obvious fλ = v relation, we can write E = hf as mv2/2 = v/λ and, hence, after moving one of the two v’s in v2 (and the 1/2 factor) on the left side to the right side of this equation, we get mv = 2h/λ. So there we are:
p = mv = 2h/λ.
Well… No. The second de Broglie relation is just p = h/λ. There is no factor 2 in it. So what’s wrong?
A factor of 2 in an equation like this surely doesn’t matter, does it? It does. We are talking tiny wavelengths here but a wavelength of 1 nanometer (1×10–9 m) – this is just an example of the scale we’re talking about here – is not the same as a wavelength of 0.5 nm. There’s another problem too. Let’s go back to our an example of an electron with a mass of 9.1×10–31 kg (that’s very tiny, and so you’ll usually see it expressed in a unit that’s more appropriate to the atomic scale), moving about with a velocity of 2.2×106 m/s (that’s the estimated speed of orbit of an electron around a hydrogen nucleus: it’s fast (2,200 km per second), but still less than 1% of the speed of light), and let’s do the math.
[Before I do the math, however, let me quickly insert a line on that ‘other unit’ to measure mass. You will usually see it written down as eV, so that’s electronvolt. Electronvolt is a measure of energy but that’s OK because mass is energy according to Einstein’s mass-energy equation: E = mc2. The point to note is that the actual measure for mass at the atomic scale is eV/c2, so we make the unit even smaller by dividing the eV (which already is a very tiny amount of energy) by c2: 1 eV/c2 corresponds to 1.782662×10−36 kg, so the mass of our electron (9.1×10–31 kg) is about 510,000 eV/c2, or 0.510 MeV/c2. I am spelling it out because you will often just see 0.510 MeV in older or more popular publications, but so don’t forget that c2 factor. As for the calculations below, I just stick to the kg and m measures because they make the dimensions come out right.]
According to our kinetic energy formula (K.E. = mv2/2), these mass and velocity values correspond to an energy value of 22 ×10−19 Joule (the Joule is the so-called SI unit for energy – don’t worry about it right now). So, from the first de Broglie equation (f = E/h) – and using the right value for Planck’s constant (6.626 J·s), we get a frequency of 3.32×1015 hertz (hertz just means oscillations per second as you know). Now, using v once again, and fλ = v, we see that corresponds to a wavelength of 0.66 nanometer (0.66×10−9 m). [Just take the numbers and do the math.]
However, if we use the second de Broglie relation, which relates wavelength to momentum instead of energy, then we get 0.33 nanometer (0.33×10−9 m), so that’s half of the value we got from the first equation. So what is it: 0.33 or 0.66 nm? It’s that factor 2 again. Something is wrong.
It must be that kinetic energy formula. You’ll say we should include potential energy or something. No. That’s not the issue. First, we’re talking a free particle here: an electron moving in space (a vacuum) with no external forces acting on it, so it’s a field-free space (or a region of constant potential). Second, we could, of course, extend the analysis and include potential energy, and show how it’s converted to kinetic energy (like a stone falling from 100 m to 50 m: potential energy gets converted into kinetic energy) but making our analysis more complicated by including potential energy as well will not solve our problem here: it will only make you even more confused.
Then it must be some relativistic effect you’ll say. No. It’s true the formula for kinetic energy above only holds for relatively low speeds (as compared to light, so ‘relatively’ low can be thousands of km per second) but that’s not the problem here: we are talking electrons moving at non-relativistic speeds indeed, so their mass or energy is not (or hardly) affected by relativistic effects and, hence, we can indeed use the more simple non-relativistic formulas.
The real problem we’re encountering here is not with the equations: it’s the simplistic model of our wave. We are imagining one wave here indeed, with a single frequency, a single wavelength and, hence, one single velocity – which happens to coincide with the velocity of our particle. Such wave cannot possibly represent an actual de Broglie wave: the wave is everywhere and, hence, the particle it represents is nowhere. Indeed, a wave defined by a specific wavelength λ (or a wave number k = 2π/λ if we’re using complex number notation) and a specific frequency f or period T (or angular frequency ω = 2π/T = 2πf) will have a very regular shape – such as Ψ = Aei(ωt-kx) and, hence, the probability of actually locating that particle at some specific point in space will be the same everywhere: |Ψ|2 = |Aei(ωt-kx)|2 = A2. [If you are confused about the math here, I am sorry but I cannot re-explain this once again: just remember that our de Broglie wave represents probability amplitudes – so that’s some complex number Ψ = Ψ(x, t) depending on space and time – and that we need to take the modulus squared of that complex number to get the probability associated with some (real) value x (i.e. the space variable) and some value t (i.e. the time variable).]
So the actual matter wave of a real-life electron will be represented by a wave train, or a wave packet as it is usually referred to. Now, a wave packet is described by (at least) two types of wave velocity:
- The so-called group velocity: the group velocity of a wave is denoted by vg and is the velocity of the wave packet as a whole is traveling. Wikipedia defines it as “the velocity with which the overall shape of the waves’ amplitudes — known as the modulation or envelope of the wave — propagates through space.”
- The so-called phase velocity: the phase velocity is denoted by vp and is what we usually associate with the velocity of a wave. It is just what it says it is: the rate at which the phase of the (composite) wave travels through space.
The term between brackets above – ‘composite’ – already indicates what it’s all about: a wave packet is to be analyzed as a composite wave: so it’s a wave composed of a finite or infinite number of component waves which all have their own wave number k and their own angular frequency ω. So the mistake we made above is that, naively, we just assumed that (i) there is only one simple wave (and, of course, there is only one wave, but it’s not a simple one: it’s a composite wave), and (ii) that the velocity v of our electron would be equal to the velocity of that wave. Now that we are a little bit more enlightened, we need to answer two questions in regard to point (ii):
- Why would that be the case?
- If it’s is the case, then what wave velocity are we talking about: the group velocity or the phase velocity?
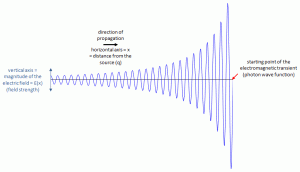
To answer both questions, we need to look at wave packets once again, so let’s do that. Just to visualize things, I’ll insert – once more – that illustration you’ve seen in my other posts already:
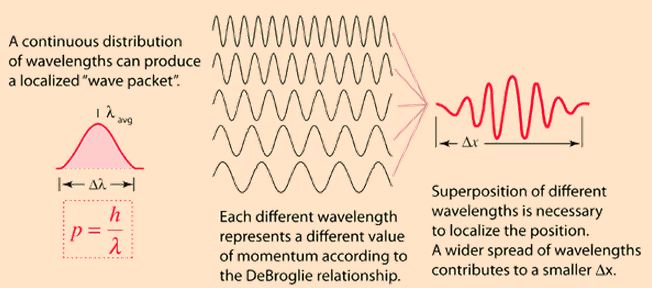
The de Broglie wave packet
The Wikipedia article on the group velocity of a wave has wonderful animations, which I would advise you to look at in order to make sure you are following me here. There are several possibilities:
- The phase velocity and the group velocity are the same: that’s a rather unexciting possibility but it’s the easiest thing to work with and, hence, most examples will assume that this is the case.
- The group and phase velocity are not the same, but our wave packet is ‘stable’, so to say. In other words, the individual peaks and troughs of the wave within the envelope travel at a different speed (the phase velocity vg), but the envelope as a whole (so the wave packet as such) does not get distorted as it travels through space.
- The wave packet dissipates: in this case, we have a constant group velocity, but the wave packet delocalizes. Its width increases over time and so the wave packet diffuses – as time goes by – over a wider and wider region of space, until it’s actually no longer there. [In case you wonder why it did not group this third possibility under (2): it’s a bit difficult to assign a fixed phase velocity to a wave like this.]
How the wave packet will behave depends on the characteristics of the component waves. To be precise, it will depend on their angular frequency and their wave number and, hence, their individual velocities. First, note the relationship between these three variables: ω = 2πf and k = 2π/λ so ω/k = fλ = v. So these variables are not independent: if you have two values (e.g. v and k), you also have the third one (ω). Secondly, note that the component waves of our wave packet will have different wavelengths and, hence, different wave numbers k.
Now, the de Broglie relation p = ħk (i.e. the same relation as p = h/λ but we replace λ with 2π/k and then ħ is the so-called reduced Planck constant ħ = h/2π) makes it obvious that different wave numbers k correspond to different values p for the momentum of our electron, so allowing for a spread in k (or a spread in λ as illustrates above) amounts to allowing for some spread in p. That’s where the uncertainty principle comes in – which I actually derived from a theoretical wave function in my post on Fourier transforms and conjugate variables. But so that’s not something I want to dwell on here.
We’re interested in the ω’s. What about them? Well… ω can take any value really – from a theoretical point of view that is. Now you’ll surely object to that from a practical point of view, because you know what it implies: different velocities of the component waves. But you can’t object in a theoretical analysis like this. The only thing we could possible impose as a constraint is that our wave packet should not dissipate – so we don’t want it to delocalize and/or vanish after a while because we’re talking about some real-life electron here, and so that’s a particle which just doesn’t vanish like that.
To impose that condition, we need to look at the so-called dispersion relation. We know that we’ll have a whole range of wave numbers k, but so what values should ω take for a wave function to be ‘well-behaved’, i.e. not disperse in our case? Let’s first accept that k is some variable, the independent variable actually, and so then we associate some ω with each of these values k. So ω becomes the dependent variable (dependent on k that is) and that amounts to saying that we have some function ω = ω(k).
What kind of function? Well… It’s called the dispersion relation – for rather obvious reasons: because this function determines how the wave packet will behave: non-dispersive or – what we don’t want here – dispersive. Indeed, there are several possibilities:
- The speed of all component waves is the same: that means that the ratio ω/k = v is the same for all component waves. Now that’s the case only if ω is directly proportional to k, with the factor of proportionality equal to v. That means that we have a very simple dispersion relation: ω = αk with α some constant equal to the velocity of the component waves as well as the group and phase velocity of the composite wave. So all velocities are just the same (v = vp = vg = α) and we’re in the first of the three cases explained at the beginning of this section.
- There is a linear relation between ω and k but no direct proportionality, so we write ω = αk + β, in which β can be anything but not some function of k. So we allow different wave speeds for the component waves. The phase velocity will, once again, be equal to the ratio of the angular frequency and the wave number of the composite wave (whatever that is), but what about the group velocity, i.e. the velocity of our electron in this example? Well… One can show – but I will not do it here because it is quite a bit of work – that the group velocity of the wave packet will be equal to vg = dω/dk, i.e. the (first-order) derivative of ω with respect to k. So, if we want that wave packet to travel at the same speed of our electron (which is what we want of course because, otherwise, the wave packet would obviously not represent our electron), we’ll have to impose that dω/dk (or ∂ω/∂k if you would want to introduce more independent variables) equals v. In short, we have the condition that dω/dk = d(αk + β)/dk = α = k.
- If the relation between ω and k is non-linear, well… Then we have none of the above. Hence, we then have a wave packet that gets distorted and stretched out and actually vanishes after a while. That case surely does not represent an electron.
Back to the de Broglie wave relations
Indeed, it’s now time to go back to our de Broglie relations – E = hf and p = h/λ and the question that sparked the presentation above: what formula to use for E? Indeed, for p it’s easy: we use p = mv and, if you want to include the case of relativistic speeds, you will write that formula in a more sophisticated way by making it explicit that the mass m is the relativistic mass m = γm0: the rest mass multiplied with a factor referred to as the Lorentz factor which, I am sure, you have seen before: γ = (1 – v2/c2)–1/2. At relativistic speeds (i.e. speeds close to c), this factor makes a difference: it adds mass to the rest mass. So the mass of a particle can be written as m = γm0, with m0 denoting the rest mass. At low speeds (e.g. 1% of the speed of light – as in the case of our electron), m will hardly differ from m0 and then we don’t need this Lorentz factor. It only comes into play at higher speeds.
At this point, I just can’t resist a small digression. It’s just to show that it’s not ‘relativistic effects’ that cause us trouble in finding the right energy equation for our E = hf relation. What’s kinetic energy? Well… There’s a few definitions – such as the energy gathered through converting potential energy – but one very useful definition in the current context is the following: kinetic energy is the excess of a particle over its rest mass energy. So when we’re looking at high-speed or high-energy particles, we will write the kinetic energy as K.E. = mc2 – m0c2 = (m – m0)c2 = γm0c2 – m0c2 = m0c2(γ – 1). Before you think I am trying to cheat you: where is the v of our particle? [To make it specific: think about our electron once again but not moving at leisure this time around: imagine it’s speeding at a velocity very close to c in some particle accelerator. Now, v is close to c but not equal to c and so it should not disappear. […]
It’s in the Lorentz factor γ = (1 – v2/c2)–1/2.
Now, we can expand γ into a binomial series (it’s basically an application of the Taylor series – but just check it online if you’re in doubt), so we can write γ as an infinite sum of the following terms: γ = 1 + (1/2)·v2/c2 + (3/8)·v4/c4 + (3/8)·v4/c4 + (5/16)·v6/c6 + … etcetera. [The binomial series is an infinite Taylor series, so it’s not to be confused with the (finite) binomial expansion.] Now, when we plug this back into our (relativistic) kinetic energy equation, we can scrap a few things (just do it) to get where I want to get:
K.E. = (1/2)·m0v2 + (3/8)·m0v4/c2 + (5/16)·m0v6/c4 + … etcetera
So what? Well… That’s it – for the digression at least: see how our non-relativistic formula for kinetic energy (K.E. = m0v2/2 is only the first term of this series and, hence, just an approximation: at low speeds, the second, third etcetera terms represent close to nothing (and more close to nothing as you check out the fourth, fifth etcetera terms). OK, OK… You’re getting tired of these games. So what? Should we use this relativistic kinetic energy formula in the de Broglie relation?
No. As mentioned above already, we don’t need any relativistic correction, but the relativistic formula above does come in handy to understand the next bit. What’s the next bit about?
Well… It turns out that we actually do have to use the total energy – including (the energy equivalent to) the rest mass of our electron – in the de Broglie relation E = hf.
WHAT!?
If you think a few seconds about the math of this – so we’d use γm0c2 instead of (1/2)m0v2 (so we use the speed of light instead of the speed of our particle) – you’ll realize we’ll be getting some astronomical frequency (we got that already but so here we are talking some kind of truly fantastic frequency) and, hence, combining that with the wavelength we’d derive from the other de Broglie equation (p = h/λ) we’d surely get some kind of totally unreal speed. Whatever it is, it will surely have nothing to do with our electron, does it?
Let’s go through the math.
The wavelength is just the same as that one given by p = h/λ, so we have λ = 0.33 nanometer. Don’t worry about this. That’s what it is indeed. Check it out online: it’s about a thousand times smaller than the wavelength of (visible) light but that’s OK. We’re talking something real here. That’s why electron microscopes can ‘see’ stuff that light microscopes can’t: their resolution is about a thousand times higher indeed.
But so when we take the first equation once again (E =hf) and calculate the frequency from f = γm0c2/h, we get an frequency f in the neighborhood of 12.34×1019 herz. So that gives a velocity of v = fλ = 4.1×1010 meter per second (m/s). But… THAT’S MORE THAN A HUNDRED TIMES THE SPEED OF LIGHT. Surely, we must have got it wrong.
We don’t. The velocity we are calculating here is the phase velocity vp of our matter wave – and IT’S REAL! More in general, it’s easy to show that this phase velocity is equal to vp = fλ = E/p = (γm0c2/h)·(h/γm0v) = c2/v. Just fill in the values for c and v (3×108 and 2.2×106 respectively and you will get the same answer.
But that’s not consistent with relativity, is it? It is: phase velocities can be (and, in fact, usually are – as evidenced by our real-life example) superluminal as they say – i.e. much higher than the speed of light. However, because they carry no information – the wave packet shape is the ‘information’, i.e. the (approximate) location of our electron – such phase velocities do not conflict with relativity theory. It’s like amplitude modulation, like AM radiowaves): the modulation of the amplitude carries the signal, not the carrier wave.
The group velocity, on the other hand, can obviously not be faster than c and, in fact, should be equal to the speed of our particle (i.e. the electron). So how do we calculate that? We don’t have any formula ω(k) here, do we? No. But we don’t need one. Indeed, we can write:
vg = ∂ω/∂k = ∂(E/ ħ)/∂(p/ ħ) = ∂E/∂p
[Do you see why we prefer the ∂ symbol instead of the d symbol now? ω is a function of k but it’s – first and foremost – a function of E, so a partial derivative sign is quite appropriate.]
So what? Well… Now you can use either the relativistic or non-relativistic relation between E and p to get a value for ∂E/∂p. Let’s take the non-relativistic one first (E = p2/2m) : ∂E/∂p = ∂(p2/2m)/∂p = p/m = v. So we get the velocity of our electron! Just like we wanted. 🙂 As for the relativistic formula (E = (p2c2 + m02c4)1/2), well… I’ll let you figure that one out yourself. [You can also find it online in case you’d be desperate.]
Wow! So there we are. That was quite something! I will let you digest this for now. It’s true I promised to ‘point out the differences between matter waves and light waves’ in my introduction but this post has been lengthy enough. I’ll save those ‘differences’ for the next post. In the meanwhile, I hope you enjoyed and – more importantly – understood this one. If you did, you’re a master! A real one! 🙂

 On the quantization of flux for superconducting loops: see, for example,
On the quantization of flux for superconducting loops: see, for example, 

 The resemblance with the standard diffusion equation (shown below) is, effectively, very obvious:
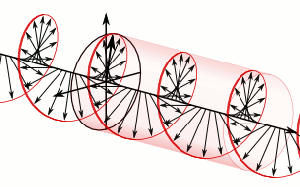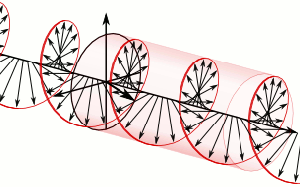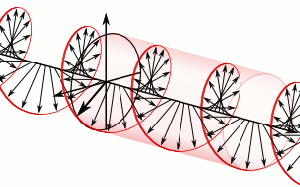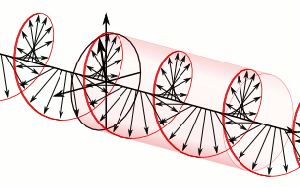
The resemblance with the standard diffusion equation (shown below) is, effectively, very obvious: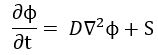 As Feynman notes, it’s just that imaginary coefficient that makes the behavior quite different. How exactly? Well… You know we get all of those complicated electron orbitals (i.e. the various wave functions that satisfy the equation) out of Schrödinger’s differential equation. We can think of these solutions as (complex) standing waves. They basically represent some equilibrium situation, and the main characteristic of each is their energy level. I won’t dwell on this because – as mentioned above – I assume you master the math. Now, you know that – if we would want to interpret these wavefunctions as something real (which is surely what I want to do!) – the real and imaginary component of a wavefunction will be perpendicular to each other. Let me copy the animation for the elementary wavefunction ψ(θ) = a·e−i∙θ = a·e−i∙(E/ħ)·t = a·cos[(E/ħ)∙t] − i·a·sin[(E/ħ)∙t] once more:
As Feynman notes, it’s just that imaginary coefficient that makes the behavior quite different. How exactly? Well… You know we get all of those complicated electron orbitals (i.e. the various wave functions that satisfy the equation) out of Schrödinger’s differential equation. We can think of these solutions as (complex) standing waves. They basically represent some equilibrium situation, and the main characteristic of each is their energy level. I won’t dwell on this because – as mentioned above – I assume you master the math. Now, you know that – if we would want to interpret these wavefunctions as something real (which is surely what I want to do!) – the real and imaginary component of a wavefunction will be perpendicular to each other. Let me copy the animation for the elementary wavefunction ψ(θ) = a·e−i∙θ = a·e−i∙(E/ħ)·t = a·cos[(E/ħ)∙t] − i·a·sin[(E/ħ)∙t] once more:



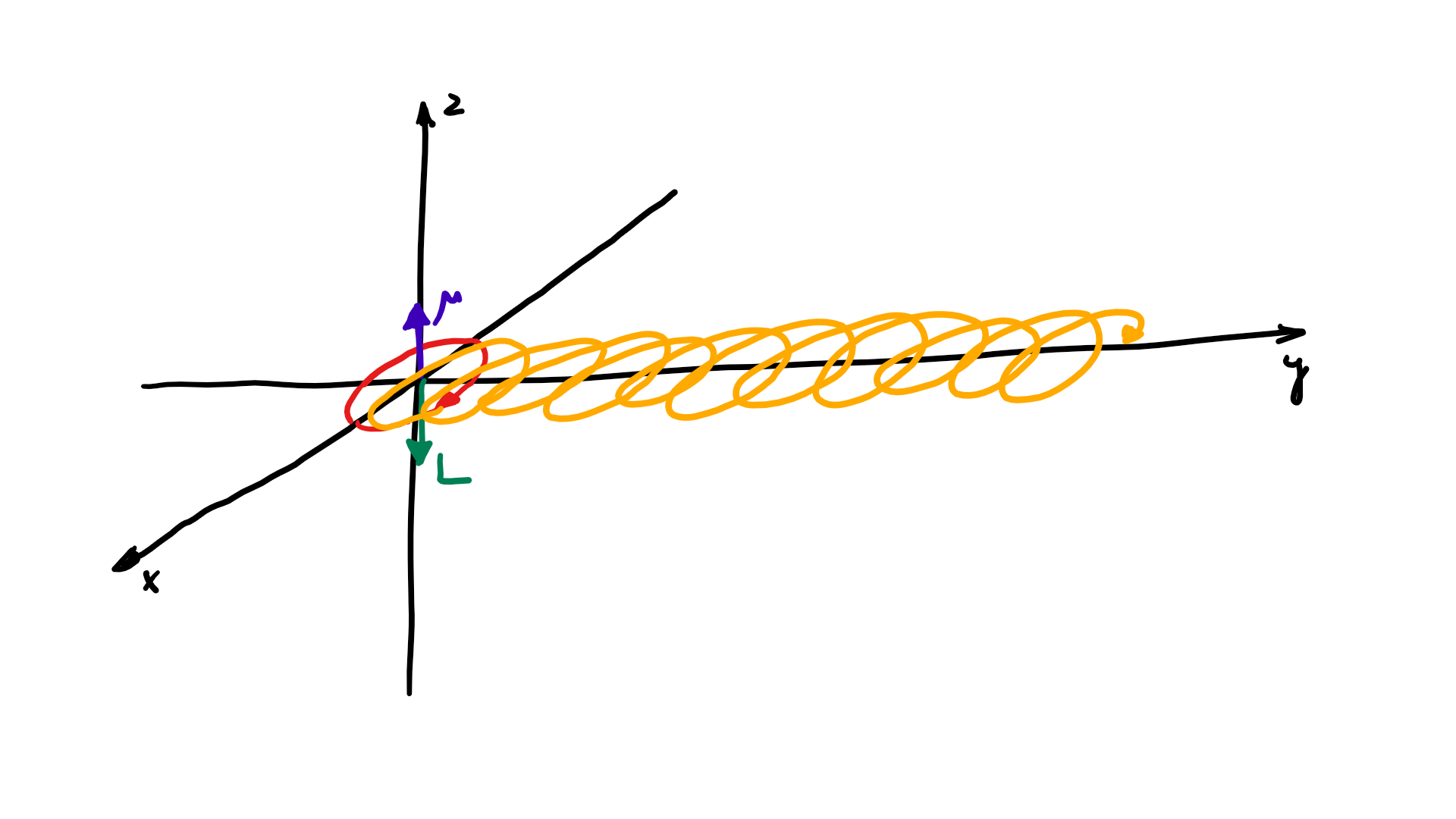 But that shouldn’t distract you. 🙂 The question here is the following: could we possibly think of a new formulation of Schrödinger’s equation – using vectors (again, real vectors – not these weird state vectors) rather than complex algebra?
But that shouldn’t distract you. 🙂 The question here is the following: could we possibly think of a new formulation of Schrödinger’s equation – using vectors (again, real vectors – not these weird state vectors) rather than complex algebra?