My previous post on the speed of light as an angular velocity was rather cryptic. This post will be a bit more elaborate. Not all that much, however: this stuff is and remains quite dense, unfortunately. 😦 But I’ll do my best to try to explain what I am thinking of. Remember the formula (or definition) of the elementary wavefunction:
ψ = a·e−i[E·t − p∙x]/ħ = a·cos(p∙x/ħ − E∙t/ħ) + i·a·sin(p∙x/ħ − E∙t/ħ)
How should we interpret this? We know an actual particle will be represented by a wave packet: a sum of wavefunctions, each with its own amplitude ak and its own argument θk = (Ek∙t − pk∙x)/ħ. But… Well… Let’s see how far we get when analyzing the elementary wavefunction itself only.
According to mathematical convention, the imaginary unit (i) is a 90° angle in the counterclockwise direction. However, Nature surely cannot be bothered about our convention of measuring phase angles – or time itself – clockwise or counterclockwise. Therefore, both right- as well as left-handed polarization may be possible, as illustrated below.
The left-handed elementary wavefunction would be written as:
ψ = a·ei[E·t − p∙x]/ħ = a·cos(p∙x/ħ − E∙t/ħ) − i·a·sin(p∙x/ħ − E∙t/ħ)
In my previous posts, I hypothesized that the two physical possibilities correspond to the angular momentum of our particle – say, an electron – being either positive or negative: J = +ħ/2 or, else, J = −ħ/2. I will come back to this in a moment. Let us first further examine the functional form of the wavefunction.
We should note that both the direction as well as the magnitude of the (linear) momentum (p) are relative: they depend on the orientation and relative velocity of our reference frame – which are, in effect, relative to the reference frame of our object. As such, the wavefunction itself is relative: another observer will obtain a different value for both the momentum (p) as well as for the energy (E). Of course, this makes us think of the relativity of the electric and magnetic field vectors (E and B) but… Well… It’s not quite the same because – as I will explain in a moment – the argument of the wavefunction, considered as a whole, is actually invariant under a Lorentz transformation.
Let me elaborate this point. If we consider the reference frame of the particle itself, then the idea of direction and momentum sort of vanishes, as the momentum vector shrinks to the origin itself: p = 0. Let us now look at how the argument of the wavefunction transforms. The E and p in the argument of the wavefunction (θ = ω∙t – k∙x = (E/ħ)∙t – (p/ħ)∙x = (E∙t – p∙x)/ħ) are, of course, the energy and momentum as measured in our frame of reference. Hence, we will want to write these quantities as E = Ev and p = pv = pv∙v. If we then use natural time and distance units (hence, the numerical value of c is equal to 1 and, hence, the (relative) velocity is then measured as a fraction of c, with a value between 0 and 1), we can relate the energy and momentum of a moving object to its energy and momentum when at rest using the following relativistic formulas:
Ev = γ·E0 and pv = γ·m0∙v = γ·E0∙v/c2
The argument of the wavefunction can then be re-written as:
θ = [γ·E0/ħ]∙t – [(γ·E0∙v/c2)/ħ]∙x = (E0/ħ)·(t − v∙x/c2)·γ = (E0/ħ)∙t’
The γ in these formulas is, of course, the Lorentz factor, and t’ is the proper time: t’ = (t − v∙x/c2)/√(1−v2/c2). Two essential points should be noted here:
1. The argument of the wavefunction is invariant. There is a primed time (t’) but there is no primed θ (θ’): θ = (Ev/ħ)·t – (pv/ħ)·x = (E0/ħ)∙t’.
2. The E0/ħ coefficient pops up as an angular frequency: E0/ħ = ω0. We may refer to it as the frequency of the elementary wavefunction.
Now, if you don’t like the concept of angular frequency, we can also write: f0 = ω0/2π = (E0/ħ)/2π = E0/h. Alternatively, and perhaps more elucidating, we get the following formula for the period of the oscillation:
T0 = 1/f0 = h/E0
This is interesting, because we can look at the period as a natural unit of time for our particle. This period is inversely proportional to the (rest) energy of the particle, and the constant of proportionality is h. Substituting E0 for m0·c2, we may also say it’s inversely proportional to the (rest) mass of the particle, with the constant of proportionality equal to h/c2. The period of an electron, for example, would be equal to about 8×10−21 s. That’s very small, and it only gets smaller for larger objects ! But what does all of this really tell us? What does it actually mean?
We can look at the sine and cosine components of the wavefunction as an oscillation in two dimensions, as illustrated below.

Look at the little green dot going around. Imagine it is some mass going around and around. Its circular motion is equivalent to the two-dimensional oscillation. Indeed, instead of saying it moves along a circle, we may also say it moves simultaneously (1) left and right and back again (the cosine) while also moving (2) up and down and back again (the sine).
Now, a mass that rotates about a fixed axis has angular momentum, which we can write as the vector cross-product L = r×p or, alternatively, as the product of an angular velocity (ω) and rotational inertia (I), aka as the moment of inertia or the angular mass: L = I·ω. [Note we write L and ω in boldface here because they are (axial) vectors. If we consider their magnitudes only, we write L = I·ω (no boldface).]
We can now do some calculations. We already know the angular velocity (ω) is equal to E0/ħ. Now, the magnitude of r in the L = r×p vector cross-product should equal the magnitude of ψ = a·e−i∙E·t/ħ, so we write: r = a. What’s next? Well… The momentum (p) is the product of a linear velocity (v) – in this case, the tangential velocity – and some mass (m): p = m·v. If we switch to scalar instead of vector quantities, then the (tangential) velocity is given by v = r·ω.
So now we only need to think about what formula we should use for the angular mass. If we’re thinking, as we are doing here, of some point mass going around some center, then the formula to use is I = m·r2. However, we may also want to think that the two-dimensional oscillation of our point mass actually describes the surface of a disk, in which case the formula for I becomes I = m·r2/2. Of course, the addition of this 1/2 factor may seem arbitrary but, as you will see, it will give us a more intuitive result. This is what we get:
L = I·ω = (m·r2/2)·(E/ħ) = (1/2)·a2·(E/c2)·(E/ħ) = a2·E2/(2·ħ·c2)
Note that our frame of reference is that of the particle itself, so we should actually write ω0, m0 and E0 instead of ω, m and E. The value of the rest energy of an electron is about 0.510 MeV, or 8.1871×10−14 N∙m. Now, this momentum should equal J = ±ħ/2. We can, therefore, derive the (Compton scattering) radius of an electron: Substituting the various constants with their numerical values, we find that a is equal 3.8616×10−13 m, which is the (reduced) Compton scattering radius of an electron. The (tangential) velocity (v) can now be calculated as being equal to v = r·ω = a·ω = [ħ·/(m·c)]·(E/ħ) = c. This is an amazing result. Let us think about it.
Substituting the various constants with their numerical values, we find that a is equal 3.8616×10−13 m, which is the (reduced) Compton scattering radius of an electron. The (tangential) velocity (v) can now be calculated as being equal to v = r·ω = a·ω = [ħ·/(m·c)]·(E/ħ) = c. This is an amazing result. Let us think about it.
In our previous posts, we introduced the metaphor of two springs or oscillators, whose energy was equal to E = m·ω2. Is this compatible with Einstein’s E = m·c2 mass-energy equivalence relation? It is. The E = m·c2 implies E/m = c2. We, therefore, can write the following:
ω = E/ħ = m·c2/ħ = m·(E/m)·/ħ ⇔ ω = E/ħ
Hence, we should actually have titled this and the previous post somewhat differently: the speed of light appears as a tangential velocity. Think of the following: the ratio of c and ω is equal to c/ω = a·ω/ω = a. Hence, the tangential and angular velocity would be the same if we’d measure distance in units of a. In other words, the radius of an electron appears as a natural distance unit here: if we’d measure ω in units of a per second, rather than in radians (which are expressed in the SI unit of distance, i.e. the meter) per second, the two concepts would coincide.
More fundamentally, we may want to look at the radius of an electron as a natural unit of velocity. Huh? Yes. Just re-write the c/ω = a as ω = c/a. What does it say? Exactly what I said, right? As such, the radius of an electron is not only a norm for measuring distance but also for time. 🙂
If you don’t quite get this, think of the following. For an electron, we get an angular frequency that is equal to ω = E/ħ = (8.19×10−14 N·m)/(1.05×10−34 N·m·s) ≈ 7.76×1020 radians per second. That’s an incredible velocity, because radians are expressed in distance units—so that’s in meter. However, our mass is not moving along the unit circle, but along a much tinier orbit. The ratio of the radius of the unit circle and a is equal to 1/a ≈ (1 m)/(3.86×10−13 m) ≈ 2.59×1012. Now, if we divide the above-mentioned velocity of 7.76×1020 radians per second by this factor, we get… Right ! The speed of light: 2.998×1082 m/s. 🙂
Post scriptum: I have no clear answer to the question as to why we should use the I = m·r2/2 formula, as opposed to the I = m·r2 formula. It ensures we get the result we want, but this 1/2 factor is actually rather enigmatic. It makes me think of the 1/2 factor in Schrödinger’s equation, which is also quite enigmatic. In my view, the 1/2 factor should not be there in Schrödinger’s equation. Electron orbitals tend to be occupied by two electrons with opposite spin. That’s why their energy levels should be twice as much. And so I’d get rid of the 1/2 factor, solve for the energy levels, and then divide them by two again. Or something like that. 🙂 But then that’s just my personal opinion or… Well… I’ve always been intrigued by the difference between the original printed edition of the Feynman Lectures and the online version, which has been edited on this point. My printed edition is the third printing, which is dated July 1966, and – on this point – it says the following:
“Don’t forget that meff has nothing to do with the real mass of an electron. It may be quite different—although in commonly used metals and semiconductors it often happens to turn out to be the same general order of magnitude, about 2 to 20 times the free-space mass of the electron.”
Two to twenty times. Not 1 or 0.5 to 20 times. No. Two times. As I’ve explained a couple of times, if we’d define a new effective mass which would be twice the old concept – so meffNEW = 2∙meffOLD – then such re-definition would not only solve a number of paradoxes and inconsistencies, but it will also justify my interpretation of energy as a two-dimensional oscillation of mass.
However, the online edition has been edited here to reflect the current knowledge about the behavior of an electron in a medium. Hence, if you click on the link above, you will read that the effective mass can be “about 0.1 to 30 times” the free-space mass of the electron. Well… This is another topic altogether, and so I’ll sign off here and let you think about it all. 🙂

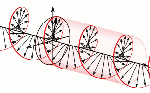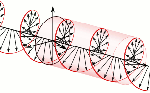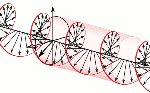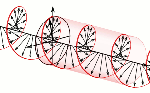
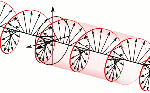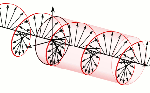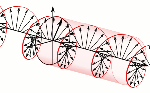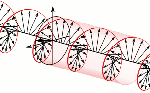
 Now, all oscillations of the elementary wavefunction have the same amplitude: a. [Terminology is a bit confusing here because we use the term amplitude to refer to two very different things here: we may say a is the amplitude of the (probability) amplitude ψ. So how many oscillations do we have? What is the size of our box? Let us assume our particle is an electron, and we will reduce its motion to a one-dimensional motion only: we’re thinking of it as traveling along the x-axis. We can then use the y- and z-axes as mathematical axes only: they will show us how the magnitude and direction of the real and imaginary component of ψ. The animation below (for which I have to credit Wikipedia) shows how it looks like.
Now, all oscillations of the elementary wavefunction have the same amplitude: a. [Terminology is a bit confusing here because we use the term amplitude to refer to two very different things here: we may say a is the amplitude of the (probability) amplitude ψ. So how many oscillations do we have? What is the size of our box? Let us assume our particle is an electron, and we will reduce its motion to a one-dimensional motion only: we’re thinking of it as traveling along the x-axis. We can then use the y- and z-axes as mathematical axes only: they will show us how the magnitude and direction of the real and imaginary component of ψ. The animation below (for which I have to credit Wikipedia) shows how it looks like. Of course, we can have right- as well as left-handed particle waves because, while time physically goes by in one direction only (we can’t reverse time), we can count it in two directions: 1, 2, 3, etcetera or −1, −2, −3, etcetera. In the latter case, think of
Of course, we can have right- as well as left-handed particle waves because, while time physically goes by in one direction only (we can’t reverse time), we can count it in two directions: 1, 2, 3, etcetera or −1, −2, −3, etcetera. In the latter case, think of 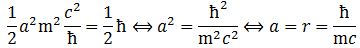 This is the formula for the radius of an electron. To be precise, it is the Compton scattering radius, so that’s the effective radius of an electron as determined by scattering experiments. You can calculate it: it is about 3.8616×10−13 m, so that’s the picometer scale, as we would expect.
This is the formula for the radius of an electron. To be precise, it is the Compton scattering radius, so that’s the effective radius of an electron as determined by scattering experiments. You can calculate it: it is about 3.8616×10−13 m, so that’s the picometer scale, as we would expect.
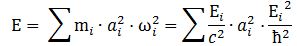

 We find that m would have to be equal to m ≈ 1.11×10−36 kg. That’s tiny. In fact, it’s equivalent to an energy of about equivalent to 0.623 eV (which you’ll see written as 623 milli-eV. This corresponds to light with a wavelength of about 2 micro-meter (μm), so that’s in the infrared spectrum. It’s a funny formula: we find, basically, that the l/a ratio is proportional to m4. Hmm… What should we think of this? If you have any ideas, let me know !
We find that m would have to be equal to m ≈ 1.11×10−36 kg. That’s tiny. In fact, it’s equivalent to an energy of about equivalent to 0.623 eV (which you’ll see written as 623 milli-eV. This corresponds to light with a wavelength of about 2 micro-meter (μm), so that’s in the infrared spectrum. It’s a funny formula: we find, basically, that the l/a ratio is proportional to m4. Hmm… What should we think of this? If you have any ideas, let me know !