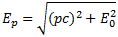Pre-script (dated 26 June 2020): Our ideas have evolved into a full-blown realistic (or classical) interpretation of all things quantum-mechanical. In addition, I note the dark force has amused himself by removing some material. So no use to read this. Read my recent papers instead. 🙂
Original post:
My previous posts were rather technical and, hence, I thought I’d re-visit a topic on which I’ve written before – but represent it from another angle: the de Broglie equation. You know it by heart: it associates a wavelength (λ) with the momentum (p) of a particle: λ = h/p. It’s a simple relationship: the wavelength and the momentum are inversely proportional, and the constant of proportionality is Planck’s constant. It’s an equation you’ll find in all of the popular accounts of quantum mechanics. However, I am of the opinion that the equation may actually not help novices to understand what quantum mechanics is all about—at least not in an initial approach.
One barrier to a proper understanding is that the de Broglie relation is always being presented as the twin of the Planck-Einstein relation for photons, which relates the energy (E) of a photon to its frequency (n): E = h∙ν = ħ∙ω [i]. It’s only natural, then, to try to relate the two equations, as momentum and energy are obviously related one to anotyher. But how exactly? What energy concept should we use? Potential energy? Kinetic energy? Should we include the equivalent energy of the rest mass?
One quickly gets into trouble here. For example, one can try the kinetic energy, K.E. = m∙v2/2 and use the definition of momentum (p = m∙v) to write E = p2/(2m), and then relate the frequency ν to the wavelength λ using the general rule that the traveling speed of a wave is equal to the product of its wavelength and its frequency (v = λ∙ν). But if E = p2/(2m) and ν = v/λ, we get:
p2/(2m) = h∙v/λ ⇔ λ = 2∙h/p
So that is almost right, but not quite: that factor 2 should not be there. In fact, it’s easy to see that we’d get de Broglie’s equation if we’d use E = m∙v2 rather than E = m∙v2/2. But E = m∙v2? How could we possibly justify the use of that formula? There’s something weird here—something deep, but I will probably die before I figure out exactly what. 🙂
Note: I should make a reference to the argument of the wavefunction here: E·t −p·x. [The argument of the wavefunction has a 1/ħ factor in front, but we assume we measure both E as well as p in units of ħ here.] So that’s an invariant quantity, i.e. it doesn’t change under a relativistic transformation of the reference frame. Now, if we measure time and distance in equivalent units, so c = 1, then we can show that E/p = 1/v. [Remember: if c would not be one, we’d write: E·β = p·c, with β = v/c, i.e. the relative velocity of our particle, as measured as a ratio of the speed of light.] As E·t − p·x, is an invariant quantity, it’s some constant that’s characteristic of the particle. But x and t change as the clock is tick. Well… Yes. But if we believe the particle is somehow real, and its velocity is v, then the ratio of the real position x and the time t should be equal to v = x/t. Hence, for these very special positions x, i.e. the real position of the particle, we can equate E·t −p·x to E·t −p·v·t = E·t −m·v·v·t = (E − m∙v2)·t. So there we have the m∙v2 factor. There must be something very deep about it, but, as mentioned above, I will probably die before I figure out exactly what. 🙂
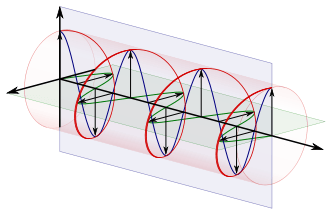
The second problem is the interpretation of λ. Of course, λ is just a length in space, which we can relate to the spatial frequency or wavenumber k = 2π/λ [ii]. And, of course, the frequency and the wavelength are, once again, related through the traveling speed of the wave: v = λ∙ν. But then, when you think about it, it’s actually not that simple: the wavefunction of a particle is much more complicated. For starters, you should think of it as a wave packet, or wavetrain, i.e. a composite wave: a sum of a potentially infinite number of elementary waves. So we do not have a simple periodic phenomenon here: we need to distinguish the so-called group velocity from the phase velocity, and we’re also talking a complex-valued wavefunction, so it’s all quite different from what we’re used to.
But back to the energy concept. We have Einstein’s E = m∙c2 = m0∙γ∙c2 equation, of course, from which the relativistically correct momentum-energy relationship can be derived [iii]:
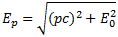
Ep is the energy of a particle with momentum p, and the relationship establishes a one-to-one relationship between the energy and the momentum of a particle [iv], with the rest mass (or rest energy) E02 = m0∙c2 appearing as a constant (the rest mass does not depend on the reference frame – per definition). However, you can try this formula too, but it will not give you the de Broglie relation. In short, it doesn’t help us in terms of understanding what the de Broglie relation is all about.
So how did this young nobleman, back in 1924, as he was writing his PhD thesis, get this λ = h/p or – using the wavenumber – the k = p/ħ equation?
Well… The relativity theory had been around for quite a while and, amongst other things (including the momentum-energy relationship above), it had also established the invariance of the four-vector product pμxμ = E∙t – p∙x = pμ‘xμ‘ = E’∙t’ – p’∙x’.
Now, any regular sinusoidal wave is associated with a phase θ = ωt – k∙x, and then quantum theory had associated a complex-valued wavefunction Ψ(θ) = Ψ(ωt – k∙x) with a particle, and so the young count, Louis de Broglie, saw the mathematical similarity between the E∙t – p∙x and ωt – k∙x expressions, and then just took the bold step of substituting ω and k for E/ħ and p/ħ respectively in the Ψ(ωt – k∙x) function. That’s it really: as the laws of physics should look the same, regardless of our frame of reference, he realized the argument of the wavefunction needed to be some invariant quantity, and so that’s what he gets through this substitution.
Of course, the substitution makes sense: it has to. To show why, let’s consider the limiting situation of a particle with zero momentum – so it’s at rest, really – but assuming that the probability of finding it at some point in space, at some point of time is equally distributed over space and time. So we have the rather nonsensical but oft-used wavefunction:
Ψ(θ) = Ψ(x, t) = a·eiθ = a·ei(ωt – k∙x)
Taking the absolute square of this yields a constant probability equal to a2 indeed. The equation is non-sensical or – to put it more politely – a limiting case only because it assumes perfect knowledge about the particle’s momentum p (we said it was zero, exactly) and, hence, about its energy. So we don’t need to worry about any Δ here: Δp = ΔE = 0 and Ep = E0. As mentioned, we know that doesn’t make much sense, and that a particle in free space will actually be represented by a wave train with a group velocity v (i.e. the classical speed of the particle) and a phase velocity, and so that’s where the modeling of uncertainty comes in, but let’s just go along with the example now. [Note that, while p = 0, that does not imply that E0 is equal to zero. Indeed, there’s energy in the rest mass and possibly potential energy too!]
Now, if we substitute ω and k for E/ħ and p/ħ respectively – note that we did away with the bold-face k and x, so we’ve reduced the analysis to one dimension (x) only – we get:
Ψ(θ) = Ψ(x, t) = a·eiθ = a·ei(E0t – p∙x)/ħ = a·ei(E0/ħ)t
What this means is that the phase θ = (E0/ħ)·t does not depend on x: it only varies in time. Hence, the diagram below – don’t look at the x’ and t’ right now: that’s another reference frame that we’ll introduce in a moment – shows equal-phase lines parallel to the x-axis and, because of the θ = (E0/ħ)·t equation, they’re equally spaced in time, i.e. in the t-coordinate.

Of course, a particle at rest in one reference frame – let’s say S – will appear to be moving in another – which we’ll denote as S’, so we have the primed coordinates x’ and t’, which are related to the x and t by the Lorentz transformation rules. It’s easy to see that the points of equal phase have a different spacing along the t’-axis, so the frequency in time must be different. Indeed, we’ll write that frequency as ω’ = Ep‘/ħ in the S’ reference frame.
Likewise, we see that the phase now does vary in space, so the probability amplitude does vary in space now, as the particle’s momentum in the primed reference frame is no longer zero: if we write it as p’, then we can write that θ = (E0/ħ)·t = (Ep‘/ħ)·t − (p/ħ)∙x = (Ep‘/ħ)·t − (p/ħ)∙x. Therefore, our wavefunction becomes:
Ψ(θ) = Ψ(x, t) = a·eiθ = a·ei(E0/ħ)t = Ψ(x’, t’) = a·ei(Ep‘·t’ − p’∙x’)/ħ
We could introduce yet another reference frame, and we’d get similar results. The point is: the k in our Ψ(θ) = Ψ(x, t) = a·eiθ = a·ei(ωt – k∙x) equation is, effectively, equal to k = p/ħ, and that identity holds in any reference frame.
Now, none of what I wrote above actually proves the de Broglie relation: it merely explains it. When everything is said and done, the de Broglie relation is a hypothesis, but it is an important one—and it does fit into the overall quantum-mechanical or wave-mechanical approach, that physicists take for granted nowadays.
So that’s it, really. I have nothing more to write but I should, perhaps, just remind you about what I said about a ‘particle wave’: we should look at it as some composite wave. Indeed, there will be uncertainty, and the uncertainty in E implies a frequency range Δω = Δ(E/ħ) = ΔE/ħ. Likewise, the momentum will be unknown, and so we’ll have a spread in the wavenumber k as well. We write: Δk = Δ(p/ħ) = Δp/ħ. We can try to reduce this uncertainty, but the Uncertainty Principle gives us the limits: the Δp·Δx and/or the ΔE·Δt products cannot be smaller than ħ/2.
So we’ll have a potentially infinite number of waves with slightly different values for ω and k, whose sum may be visualized as a complex-valued traveling wavetrain, like the lump below.

All of the component waves necessarily need to travel at the same speed, and this speed, which is the ratio of ω and k, will be equal to the so-called phase velocity of the wave (vp), which we can calculate as vp = ω/k = (E/ħ)/(p/ħ) = E/p = (m·c2)/(m·v) = c2/v. This speed is superluminal (c2/v = c/β, with β = v/c < 1), but that is not in contradiction with special relativity because the phase velocity carries no ‘signal’ or ‘information’. As for the group velocity (vg), we can effectively see that this is equal to the classical velocity of our ‘particle’ by noting that:
vg = dω/dk = d(E/ħ)/d(p/ħ) = dE/dp = d[(p2/(2m)]/dp = p/m = v.
You may think there’s some cheating here, as we equate E with the kinetic energy only. You’re right: the total energy should also include potential energy and rest energy, so E is a sum, but then rest energy and potential energy are treated as constants and, hence, it’s only the kinetic energy that matters when taking the derivative with respect to p, and so that’s why get the result we get, which makes perfect sense.
I wanted this to be a very short post, and so I will effectively end it here. I hope you enjoyed it. It actually sets the stage for a more interesting discussion, and that’s a discussion on how a change in potential energy effectively changes the phase and, hence, the amplitude. But so that’s for next time. I also need to devote a separate post on a discussion of the wave-mechanical framework in general, with a particular focus on the math behind. So… Well… Yes, the next posts are likely to be somewhat more technical again. 
[i] I should make a note on notations here, and also insert some definitions. I will denote a frequency by ν (nu), rather than by f, so as to not cause confusion with any function f. A frequency is expressed in cycles per second, while the angular frequency ω is expressed in radians per second. One cycle covers 2π radians and, therefore, we can write: ν = ω/2π. Hence, h∙ν = h∙ω/2π = ħ∙ω. Both ν as well as ω measure the time-rate of change of the phase, as opposed to k, i.e. the spatial frequency of the wave, which depends on the speed of wave. I will also use the symbol v for the speed of a wave, although that is hugely confusing, because I will also use it to denote the classical velocity of the particle. However, I find the conventional use of the symbol of c even more confusing, because this symbol is also used for the speed of light, and the speed of a wave is not necessarily equal to the speed of light. In fact, both the group as well as the phase velocity of a particle wave are very different from the speed of light. The speed of a wave and the speed of light only coincide for electromagnetic waves and, even then, it should be noted that photons also have amplitudes to travel faster or slower than the speed of light.
[ii] Note that the de Broglie relation can be re-written as k = p/ħ, with ħ = h/2π. Indeed, λ = 2π/k and, hence, we get: λ = h/p ⇔ 2π/k = h/p ⇔ p = ħ∙k ⇔ k = p/ħ.
[iii] One gets the equation by equating m to m = m0γ (γ is the Lorentz factor) in the E2 = (mc2)2 equation, and re-arranging.
[iv] Note that this energy formula does not include any potential energy: it is (equivalent) rest mass plus kinetic energy only.
Some content on this page was disabled on June 16, 2020 as a result of a DMCA takedown notice from The California Institute of Technology. You can learn more about the DMCA here:
https://wordpress.com/support/copyright-and-the-dmca/